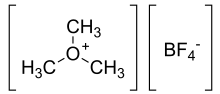
Trimethyloxonium tetrafluoroborate
| |

| |
| Names | |
|---|---|
| IUPAC name Trimethyloxonium tetrafluoroborate | |
| Other names Meerwein's salt Trimethyloxonium fluoroborate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.360 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| [(CH3)3O]+[BF4]− | |
| Molar mass | 147.91 g·mol−1 |
| Appearance | White solid |
| Melting point | 179.6–180 °C (355.3–356.0 °F; 452.8–453.1 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Trimethyloxonium tetrafluoroborate is the organic compound with the formula [(CH3)3O]+[BF4]−. (It is sometimes called "Meerwein's salt" after Hans Meerwein.[1][a]) This salt is a strong methylating agent, being a synthetic equivalent of CH+3. It is a white solid that rapidly decomposes upon exposure to atmospheric moisture, although it is robust enough to be weighed quickly without inert atmosphere protection. Triethyloxonium tetrafluoroborate is a closely related compound.
Preparation and reactions
The compound is prepared by the reaction of boron trifluoride with dimethyl ether and epichlorohydrin:[1]
- 4 Me2O·BF3 + 2 Me2O + 3 C2H3OCH2Cl → 3 [Me3O]+[BF4]− + B[(OCH(CH2Cl)CH2OMe]3
The salt hydrolyzes readily:
- [Me3O]+[BF4]− + H2O → Me2O + MeOH + H+[BF4]−
Trimethyloxonium tetrafluoroborate is generally ranked as the strongest commercially available reagent for electrophilic methylation,[1] being stronger than methyl sulfonate esters, including methyl triflate and methyl fluorosulfonate ("magic methyl").[2] Only the exotic dimethylhalonium reagents ([Me2X]+[SbF6]−, X = Cl, Br, I), methyl carboranate reagents, and the transiently-generated methyldiazonium cation (MeN+2) are stronger sources of electrophilic methyl.
Due to its high reactivity, it is rapidly destroyed by atmospheric moisture and best stored in an inert atmosphere glovebox at −20 °C. Its degradation products are corrosive, although it is considerably less hazardous than methyl triflate or methyl fluorosulfonate, on account of its lack of volatility.
Trimethyloxonium tetrafluoroborate is useful for esterification of carboxylic acids under conditions where acid-catalyzed reactions are infeasible: [3]
- RCO2H + (CH3)3OBF4 → RCO2CH3 + (CH3)2O + HBF4
References
- ^ a b c T. J. Curphey (1971). "Trimethyloxonium Tetrafluoroborate". Organic Syntheses. 51: 142. doi:10.15227/orgsyn.051.0142.
- ^ Stang, Peter J.; Hanack, Michael; Subramanian, L. R. (1982). "Perfluoroalkanesulfonic Esters: Methods of Preparation and Applications in Organic Chemistry". Synthesis. 1982 (2): 85–126. doi:10.1055/s-1982-29711. ISSN 0039-7881. S2CID 94894040.
- ^ Raber, Douglas J.; Gariano, Jr, Patrick; Brod, Albert O.; Gariano, Anne L.; Guida, Wayne C. (1977). "Esterification of Carboxylic Acids with Trialkyloxonium Salts: Ethyl and Methyl 4-Acetoxybenzoates". Organic Syntheses. 56: 59. doi:10.15227/orgsyn.056.0059.
Notes
- ^ Meerwein's salt classically referred to triethyloxonium tetrafluoroborate. However, in recent years, the trimethyloxonium salt has also been called Meerwein's salt.
