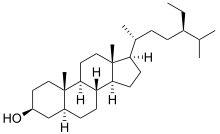
Stigmastanol
| |
| Names | |
|---|---|
| IUPAC name 5α-Stigmastan-3β-ol | |
| Systematic IUPAC name (1R,3aS,3bR,5aS,7S,9aS,9bS,11aR)-1-[(2R,5R)-5-Ethyl-6-methylheptan-2-yl]-9a,11a-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-7-ol | |
| Other names (3β)-Stigmastan-3-ol; (3β,5α)-Stigmastan-3-ol; β-Sitostanol; Dihydrositosterin; Dihydrositosterol; Dihydro-β-sitosterol; Fucostanol; Spinastanol; 24α-Ethylcholestanol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.345 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C29H52O | |
| Molar mass | 416.734 g·mol−1 |
| Boiling point | 139.4 to 139.8 °C (282.9 to 283.6 °F; 412.5 to 412.9 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Stigmastanol (sitostanol) is a phytosterol found in a variety of plant sources. Similar to sterol esters and stanol esters, stigmastanol inhibits the absorption of cholesterol from the diet.[2][3][4] Animal studies suggest that it also inhibits biosynthesis of cholesterol in the liver.[2][verification needed]
Stigmastanol is the product of the reduction of β-sitosterol[5] and the hydrogenation of stigmasterol.[6]
See also
- Stigmasterol, a closely related sterol
References
- ^ Sandqvist, Hakan; Bengtsson, Edvard (1931). "The empirical formula of sitosterol". Berichte der Deutschen Chemischen Gesellschaft B. 64: 2167–2171. doi:10.1002/cber.19310640849.
- ^ a b Batta, Ashok K.; Xu, Guorong; Honda, Akira; Miyazaki, Teruo; Salen, Gerald (2006). "Stigmasterol reduces plasma cholesterol levels and inhibits hepatic synthesis and intestinal absorption in the rat". Metabolism: Clinical and Experimental. 55 (3): 292–299. doi:10.1016/j.metabol.2005.08.024. PMID 16483871.
- ^ Heinemann T, Pietruck B, Kullak-Ublick G, von Bergmann K (1988). "Comparison of sitosterol and sitostanol on inhibition of intestinal cholesterol absorption". Agents and Actions. Supplements. 26: 117–122. PMID 3265272.
- ^ Heinemann T, Kullak-Ublick GA, Pietruck B, von Bergmann K (1991). "Mechanisms of action of plant sterols on inhibition of cholesterol absorption. Comparison of sitosterol and sitostanol". European Journal of Clinical Pharmacology. 40 (Suppl 1): S59–63. doi:10.1007/BF01409411. PMID 2044646. S2CID 23279253.
- ^ Thomasson Crompton, David William; Nickol, Brent B. (1985). Biology of the Acanthocephala. Cambridge University Press. p. 185. ISBN 9780521246743.
- ^ Paxena, P. B. (2007). Chemistry of Alkaloids. Discovery Publishing Hous. p. 231. ISBN 9788183563161.
