Walden inversion
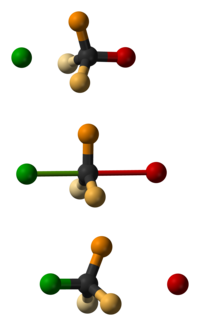
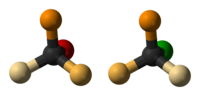
Walden inversion is the inversion of a stereogenic center in a chiral molecule in a chemical reaction. Since a molecule can form two enantiomers around a stereogenic center, the Walden inversion converts the configuration of the molecule from one enantiomeric form to the other. For example, in an SN2 reaction, Walden inversion occurs at a tetrahedral carbon atom. It can be visualized by imagining an umbrella turned inside-out in a gale. In the Walden inversion, the backside attack by the nucleophile in an SN2 reaction gives rise to a product whose configuration is opposite to the reactant. Therefore, during SN2 reaction, 100% inversion of product takes place. This is known as Walden inversion.
It was first observed by chemist Paul Walden in 1896. He was able to convert one enantiomer of a chemical compound into the other enantiomer and back again in a so-called Walden cycle which went like this: (+) chlorosuccinic acid (1 in the illustration) was converted to (+) malic acid 2 by action of silver oxide in water with retention of configuration. In the next step the hydroxyl group was replaced by chlorine to the other isomer of chlorosuccinic acid 3 by reaction with phosphorus pentachloride. A reaction with silver oxide yielded (-) malic acid 4 and finally a reaction with PCl5 returned the cycle to its starting point.[1]
In this reaction, the silver oxide in the first step acts as a hydroxide donor while the silver ion plays no role in the reaction. The intermediates are the carboxyl dianion A which gives an intramolecular nucleophilic substitution by the β-carboxylate anion to produce a four-membered β-lactone ring B. The α-carboxyl group is also reactive but in silico data suggests that the transition state for the formation of the three-membered α-lactone is very high. A hydroxyde ion ring-opens the lactone to form the alcohol C and the net effect of two counts of inversion is retention of configuration.[2]
See also
- Another demonstration of the Walden cycle in the Brook rearrangement.
References
- ^ P. Walden (1896). "Ueber die gegenseitige Umwandlung optischer Antipoden". Berichte der deutschen chemischen Gesellschaft. 29 (1): 133–138. doi:10.1002/cber.18960290127.
- ^ Buchanan, J. Grant; Diggle, Richard A.; Ruggiero, Giuseppe D.; Williams, Ian H. (2006). "The Walden cycle revisited: a computational study of competitive ring closure to α- and β-lactones". Chemical Communications (10). Royal Society of Chemistry (RSC): 1106. doi:10.1039/b517461a. ISSN 1359-7345.

