Sitravatinib
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
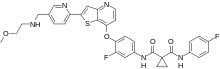
| Formula | C33H29F2N5O4S |
| Molar mass | 629.68 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Sitravatinib (MGCD516) is an experimental drug for the treatment of cancer. It is a small molecule inhibitor of multiple tyrosine kinases.[1]
Sitravatinib is being developed by Mirati Therapeutics.[2]
Ongoing phase II trials include a trial for liposarcoma,[3] a combination trial for non-small cell lung cancer,[4] and a combination trial with nivolumab for renal cell carcinoma.[5] Sitravatinib is being evaluated in ongoing trials in patients with advanced non-small cell lung cancer, including in a combination trial with nivolumab in those who are resistant to checkpoint inhibitor therapy, and certain patients who are naïve to checkpoint inhibitor therapy.[6]
References
- ^ Yang Y, Ji N, Cai CY, Wang JQ, Lei ZN, Teng QX, Wu ZX, Cui Q, Pan Y, Chen ZS (July 2020). "Modulating the function of ABCB1: in vitro and in vivo characterization of sitravatinib, a tyrosine kinase inhibitor". Cancer Communications. 40 (7): 285–300. doi:10.1002/cac2.12040. PMC 7365458. PMID 32525624.
- ^ "Sitravatinib | Mirati Therapeutics, Inc".
- ^ Clinical trial number NCT02978859 for "MGCD516 in Advanced Liposarcoma and Other Soft Tissue Sarcomas" at ClinicalTrials.gov
- ^ Clinical trial number NCT02954991 for "Phase 2 Study of Glesatinib, Sitravatinib or Mocetinostat in Combination With Nivolumab in Non-Small Cell Lung Cancer" at ClinicalTrials.gov
- ^ Clinical trial number NCT03015740 for "MGCD516 Combined With Nivolumab in Renal Cell Cancer (RCC)" at ClinicalTrials.gov
- ^ "A Randomized Phase 3 Study of Sitravatinib in Combination with Nivolumab Versus Docetaxel in Patients with Advanced Non-Squamous Non-Small Cell Lung Cancer with Disease Progression on or After Platinum-Based Chemotherapy and Checkpoint Inhibitor Therapy SAPPHIRE". 28 February 2022.
