Receptor activated solely by a synthetic ligand
A receptor activated solely by a synthetic ligand (RASSL) or designer receptor exclusively activated by designer drugs (DREADD), is a class of artificially engineered protein receptors used in the field of chemogenetics which are selectively activated by certain ligands.[1] They are used in biomedical research, in particular in neuroscience to manipulate the activity of neurons.[2]
Originally differentiated by the approach used to engineer them, RASSLs and DREADDs are often used interchangeably now to represent an engineered receptor-ligand system.[3] These systems typically utilize G protein-coupled receptors (GPCR) engineered to respond exclusively to synthetic ligands, like clozapine N-oxide (CNO),[4] and not to endogenous ligands. Several types of these receptors exists, derived from muscarinic or κ-opioid receptors.[1]
Types of RASSLs / DREADDs
One of the first DREADDs was based on the human M3 muscarinic receptor (hM3).[5] Only two point mutations of hM3 were required to achieve a mutant receptor with nanomolar potency for CNO, insensitivity to acetylcholine and low constitutive activity and this DREADD receptor was named hM3Dq. M1 and M5 muscarinic receptors have been mutated to create DREADDs hM1Dq and hM5Dq respectively.[5]
The most commonly used inhibitory DREADD is hM4Di, derived from the M4 muscarinic receptor that couples with the Gi protein.[5] Another Gi coupled human muscarinic receptor, M2, was also mutated to obtain the DREADD receptor hM2D.[5] Another inhibitory Gi-DREADD is the kappa-opioid-receptor (KOR) DREADD (KORD) which is selectively activated by salvinorin B (SalB).[6]
Gs-coupled DREADDs have also been developed. These receptors are also known as GsD and are chimeric receptors containing intracellular regions of the turkey erythrocyte β-adrenergic receptor substituted into the rat M3 DREADD.[7]
RASSL / DREADD ligands
A growing number of ligands that can be used to activate RASSLs / DREADDs are commercially available.[8]
CNO is the prototypical DREADD activator. CNO activates the excitatory Gq- coupled DREADDs: hM3Dq, hM1Dq and hM5Dq and also the inhibitory hM4Di and hM2Di Gi-coupled DREADDs. CNO also activates the Gs-coupled DREADD (GsD) and the β-arrestin preferring DREADD: rM3Darr (Rq(R165L).[9]
Recent findings suggest that systemically administered CNO does not readily cross the blood-brain-barrier in vivo and converts to clozapine which itself activates DREADDs. Clozapine is an atypical antipsychotic which has been indicated to show high DREADD affinity and potency. Subthreshold injections of clozapine itself can be utilised to induce preferential DREADD-mediated behaviors. Therefore, when using CNO, care must be taken in experimental design and proper controls should be incorporated.[10]
DREADD agonist 21, also known as Compound 21, represents an alternative agonist for muscarinic-based DREADDs and an alternative to CNO. It has been reported that Compound 21 has excellent bioavailability, pharmacokinetic properties and brain penetrability and does not undergo reverse metabolism to clozapine.[11] Another known agonist is perlapine, a hypnotic drug approved for treating insomnia in Japan. It acts as an activator of Gq-, Gi-, and Gs DREADDs that has structural similarity to CNO.[12] A more recent agonist of hM3Dq and hM4Di is deschloroclozapine (DCZ).[13]
On the other hand, SalB B is a potent and selective activator of KORD.[14]
JHU37160 and JHU37152 have been marketed commercially as novel DREADD ligands, active in vivo, with high potency and affinity for hM3Dq and hM4Di DREADDs.[citation needed]
Dihydrochloride salts of DREADD ligands that are water-soluble (but with differing stabilities in solution) have also been commercially developed (see [15][16] for aqueous stability).
Mechanism
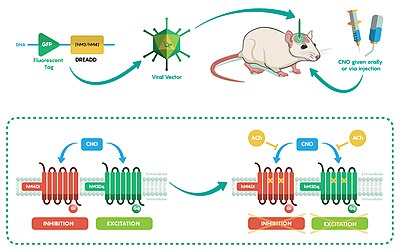
RASSLs and DREADDs are families of designer G-protein-coupled receptors (GPCRs) built specifically to allow for precise spatiotemporal control of GPCR signaling in vivo. These engineered GPCRs are unresponsive to endogenous ligands but can be activated by nanomolar concentrations of pharmacologically inert, drug-like small molecules. Currently, RASSLs exist for the interrogation of several GPCR signaling pathways, including those activated by Gs, Gi, Gq, Golf and β-arrestin.[18] A major cause for success of RASSL resources has been open exchange of DNA constructs, and RASSL related resources.
The hM4Di-DREADD's inhibitory effects are a result of the CNO's stimulation and resulting activation of the G-protein inwardly rectifying potassium (GIRK) channels. This causes hyperpolarization of the targeted neuronal cell and thus attenuates subsequent activity.[19]
Uses
This chemogenetic technique can be used for remote manipulation of cells, in particular excitable cells like neurons, both in vitro and in vivo with the administration of specific ligands.[2] Similar techniques in this field include thermogenetics and optogenetics, the control of neurons with temperature or light, respectively.[2]
Viral expression of DREADD proteins, both in-vivo enhancers and inhibitors of neuronal function, have been used to bidirectionally control behaviors in mice (e.g odor discrimination).[20] Due to their ability to modulate neuronal activity, DREADDs are used as a tool to evaluate both the neuronal pathways and behaviors associated with drug-cues and drug addiction.[21]
History
Strader and colleagues designed the first GPCR which could be activated only by a synthetic compound[22] and has gradually been gaining momentum. The first international RASSL meeting was scheduled for April 6, 2006. A simple example of the use of a RASSL system in behavioral genetics was illustrated by Mueller et al. (2005) where they showed that expressing a RASSL receptor in sweet taste cells of the mouse tongue led to a strong preference for oral consumption of the synthetic ligand, whereas expressing the RASSL in bitter taste cells caused dramatic taste aversion for the same compound.[23]
The attenuating effects of the hM4Di-DREADD were originally explored in 2007, before being confirmed in 2014.[19]
References
- ^ a b Roth BL (February 2016). "DREADDs for Neuroscientists". Neuron. 89 (4): 683–94. doi:10.1016/j.neuron.2016.01.040. PMC 4759656. PMID 26889809.
- ^ a b c Campbell, Erin J.; Marchant, Nathan J. (April 2018). "The use of chemogenetics in behavioural neuroscience: receptor variants, targeting approaches and caveats". British Journal of Pharmacology. 175 (7): 994–1003. doi:10.1111/bph.14146. ISSN 1476-5381. PMC 5843707. PMID 29338070.
- ^ Conklin BR, Hsiao EC, Claeysen S, Dumuis A, Srinivasan S, Forsayeth JR, Guettier JM, Chang WC, Pei Y, McCarthy KD, Nissenson RA, Wess J, Bockaert J, Roth BL (August 2008). "Engineering GPCR signaling pathways with RASSLs". Nature Methods. 5 (8): 673–8. doi:10.1038/nmeth.1232. PMC 2703467. PMID 18668035.
- ^ Armbruster BN, Li X, Pausch MH, Herlitze S, Roth BL (March 2007). "Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand". Proceedings of the National Academy of Sciences of the United States of America. 104 (12): 5163–8. Bibcode:2007PNAS..104.5163A. doi:10.1073/pnas.0700293104. PMC 1829280. PMID 17360345.
- ^ a b c d Armbruster, Blaine N.; Li, Xiang; Pausch, Mark H.; Herlitze, Stefan; Roth, Bryan L. (2007-03-20). "Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand". Proceedings of the National Academy of Sciences of the United States of America. 104 (12): 5163–5168. Bibcode:2007PNAS..104.5163A. doi:10.1073/pnas.0700293104. ISSN 0027-8424. PMC 1829280. PMID 17360345.
- ^ Vardy, Eyal; Robinson, J. Elliott; Li, Chia; Olsen, Reid H. J.; DiBerto, Jeffrey F.; Giguere, Patrick M.; Sassano, Flori M.; Huang, Xi-Ping; Zhu, Hu (2015-05-20). "A New DREADD Facilitates the Multiplexed Chemogenetic Interrogation of Behavior". Neuron. 86 (4): 936–946. doi:10.1016/j.neuron.2015.03.065. ISSN 1097-4199. PMC 4441592. PMID 25937170.
- ^ Conklin, Bruce R.; Hsiao, Edward C.; Claeysen, Sylvie; Dumuis, Aline; Srinivasan, Supriya; Forsayeth, John R.; Guettier, Jean-Marc; Chang, W. C.; Pei, Ying (August 2008). "Engineering GPCR signaling pathways with RASSLs". Nature Methods. 5 (8): 673–678. doi:10.1038/nmeth.1232. ISSN 1548-7105. PMC 2703467. PMID 18668035.
- ^ "DREADD ligands (CNO / SalB) for neuroscience research". HelloBio.com. Retrieved 2018-12-05.
- ^ Wess, Jürgen; Nakajima, Kenichiro; Jain, Shalini (July 2013). "Novel designer receptors to probe GPCR signaling and physiology". Trends in Pharmacological Sciences. 34 (7): 385–392. doi:10.1016/j.tips.2013.04.006. ISSN 1873-3735. PMC 3758874. PMID 23769625.
- ^ Gomez, Juan L.; Bonaventura, Jordi; Lesniak, Wojciech; Mathews, William B.; Sysa-Shah, Polina; Rodriguez, Lionel A.; Ellis, Randall J.; Richie, Christopher T.; Harvey, Brandon K. (August 4, 2017). "Chemogenetics revealed: DREADD occupancy and activation via converted clozapine". Science. 357 (6350): 503–507. Bibcode:2017Sci...357..503G. doi:10.1126/science.aan2475. ISSN 1095-9203. PMC 7309169. PMID 28774929.
- ^ Thompson, Karen J.; Khajehali, Elham; Bradley, Sophie J.; Navarrete, Jovana S.; Huang, Xi Ping; Slocum, Samuel; Jin, Jian; Liu, Jing; Xiong, Yan (2018-09-14). "DREADD Agonist 21 Is an Effective Agonist for Muscarinic-Based DREADDs in Vitro and in Vivo". ACS Pharmacology & Translational Science. 1 (1): 61–72. doi:10.1021/acsptsci.8b00012. PMC 6407913. PMID 30868140.
- ^ Chen, Xin; Choo, Hyunah; Huang, Xi-Ping; Yang, Xiaobao; Stone, Orrin; Roth, Bryan L.; Jin, Jian (2015-03-18). "The first structure-activity relationship studies for designer receptors exclusively activated by designer drugs". ACS Chemical Neuroscience. 6 (3): 476–484. doi:10.1021/cn500325v. ISSN 1948-7193. PMC 4368042. PMID 25587888.
- ^ Nagai, Yuji; Miyakawa, Naohisa; Takuwa, Hiroyuki; Hori, Yukiko; Oyama, Kei; Ji, Bin; Takahashi, Manami; Huang, Xi-Ping; Slocum, Samuel T.; DiBerto, Jeffrey F.; Xiong, Yan (September 2020). "Deschloroclozapine, a potent and selective chemogenetic actuator enables rapid neuronal and behavioral modulations in mice and monkeys". Nature Neuroscience. 23 (9): 1157–1167. doi:10.1038/s41593-020-0661-3. ISSN 1546-1726. PMID 32632286. S2CID 220375204.
- ^ Marchant, Nathan J.; Whitaker, Leslie R.; Bossert, Jennifer M.; Harvey, Brandon K.; Hope, Bruce T.; Kaganovsky, Konstantin; Adhikary, Sweta; Prisinzano, Thomas E.; Vardy, Eyal (January 2016). "Behavioral and Physiological Effects of a Novel Kappa-Opioid Receptor-Based DREADD in Rats". Neuropsychopharmacology. 41 (2): 402–409. doi:10.1038/npp.2015.149. ISSN 1740-634X. PMC 5130116. PMID 26019014.
- ^ "Stability of Water-Soluble DREADD Ligands in Solution - A Technical Review". HelloBio.com. Retrieved 2018-12-05.
- ^ "Clozapine N-oxide (CNO) freebase stability and storage". HelloBio.com. Retrieved 2018-12-05.
- ^
 This article incorporates text available under the CC BY 4.0 license. Ju, William (November 1, 2023). Neuroscience. Toronto: University of Toronto. 3.4 Chemogenic methods to examine the brain behaviour.
This article incorporates text available under the CC BY 4.0 license. Ju, William (November 1, 2023). Neuroscience. Toronto: University of Toronto. 3.4 Chemogenic methods to examine the brain behaviour.
- ^ Urban DJ, Roth BL (2015-01-06). "DREADDs (designer receptors exclusively activated by designer drugs): chemogenetic tools with therapeutic utility". Annual Review of Pharmacology and Toxicology. 55 (1): 399–417. doi:10.1146/annurev-pharmtox-010814-124803. PMID 25292433.
- ^ a b Zhu H, Roth BL (May 2014). "Silencing synapses with DREADDs". Neuron. 82 (4): 723–5. doi:10.1016/j.neuron.2014.05.002. PMC 4109642. PMID 24853931.
- ^ Smith RS, Hu R, DeSouza A, Eberly CL, Krahe K, Chan W, Araneda RC (July 2015). "Differential Muscarinic Modulation in the Olfactory Bulb". The Journal of Neuroscience. 35 (30): 10773–85. doi:10.1523/JNEUROSCI.0099-15.2015. PMC 4518052. PMID 26224860.
- ^ Ferguson SM, Eskenazi D, Ishikawa M, Wanat MJ, Phillips PE, Dong Y, Roth BL, Neumaier JF (January 2011). "Transient neuronal inhibition reveals opposing roles of indirect and direct pathways in sensitization". Nature Neuroscience. 14 (1): 22–4. doi:10.1038/nn.2703. PMC 3058296. PMID 21131952.
- ^ Coward P, Wada HG, Falk MS, Chan SD, Meng F, Akil H, Conklin BR (January 1998). "Controlling signaling with a specifically designed Gi-coupled receptor". Proceedings of the National Academy of Sciences of the United States of America. 95 (1): 352–7. Bibcode:1998PNAS...95..352C. doi:10.1073/pnas.95.1.352. JSTOR 44466. PMC 18222. PMID 9419379.
- ^ Mueller KL, Hoon MA, Erlenbach I, Chandrashekar J, Zuker CS, Ryba NJ (March 2005). "The receptors and coding logic for bitter taste". Nature. 434 (7030): 225–9. Bibcode:2005Natur.434..225M. doi:10.1038/nature03352. PMID 15759003. S2CID 4383273.
Further reading
- Conklin BR, Hsiao EC, Claeysen S, Dumuis A, Srinivasan S, Forsayeth JR, et al. (August 2008). "Engineering GPCR signaling pathways with RASSLs". Nature Methods. 5 (8): 673–8. doi:10.1038/nmeth.1232. PMC 2703467. PMID 18668035.
