Lemborexant
 | |
| Clinical data | |
|---|---|
| Trade names | Dayvigo |
| Other names | E-2006 |
| License data |
|
| Pregnancy category | |
| Routes of administration | By mouth[3] |
| Drug class | Orexin receptor antagonist; Hypnotic; Sedative |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Good (≥87%)[5][6] |
| Protein binding | 94%[3] |
| Metabolism | Liver (major: CYP3A4, minor: CYP3A5)[3] |
| Metabolites | M10[3] |
| Elimination half-life | 17–19 hours or 55 hours[3][7] |
| Excretion | Feces: 57.4%[3] Urine: 29.1%[3] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C22H20F2N4O2 |
| Molar mass | 410.425 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lemborexant, sold under the brand name Dayvigo, is an orexin antagonist medication which is used in the treatment of insomnia.[3][8] It is indicated specifically for the treatment of insomnia characterized by difficulties with sleep onset and/or maintenance in adults.[3][8] The medication is taken by mouth.[3][8]
Side effects of lemborexant include somnolence, fatigue, headache, and abnormal dreams.[3][8] The medication is a dual orexin receptor antagonist (DORA).[3][8] It acts as a selective dual antagonist of the orexin receptors OX1 and OX2.[3][8] Lemborexant has a long elimination half-life of 17 to 55 hours and a time to peak of about 1 to 3 hours.[3][8] It is not a benzodiazepine or Z-drug and does not interact with GABA receptors, instead having a distinct mechanism of action.[3][8]
Lemborexant was approved for medical use in the United States in December 2019.[9][10][11] It is a schedule IV controlled substance in the United States and may have a low potential for misuse.[3][8] Besides lemborexant, other orexin receptor antagonists including suvorexant and daridorexant have also been introduced.[12][13]
Medical uses
Lemborexant is used in the treatment of insomnia in adults.[3]
A major systematic review and network meta-analysis of medications for the treatment of insomnia published in 2022 found that lemborexant had an effect size (standardized mean difference (SMD)) against placebo for treatment of insomnia at 4 weeks of 0.36 (95% CI 0.08 to 0.63) and at 3 months of 0.41 (95% CI 0.04 to 0.78).[14] Lemborexant had similar effect sizes at 4 weeks as the other evaluated and marketed orexin receptor antagonists suvorexant (SMD 0.31, 95% CI 0.01 to 0.62) and daridorexant (SMD 0.23, 95% CI –0.01 to 0.48), whereas benzodiazepines and Z-drugs generally showed larger effect sizes (e.g., SMDs of 0.45 to 0.83) than lemborexant and the other orexin receptor antagonists.[14] However, the review concluded that lemborexant and eszopiclone among all of the insomnia medications assessed had the best profiles overall in terms of efficacy, tolerability, and acceptability.[14]
Compared to benzodiazepines, there is a low risk of developing tolerance and dependence.[15] Memory and attention are not affected the next morning when taking lemborexant.[16]
Available forms
Lemborexant is available in the form of 5 and 10 mg oral film-coated tablets.[3]
Side effects
Side effects of lemborexant include somnolence or fatigue (combined preferred terms of somnolence, lethargy, fatigue, and sluggishness) (6.9% at 5 mg and 9.6% at 10 mg vs. 1.3% for placebo), headache (5.9% at 5 mg and 4.5% at 10 mg vs. 3.4% for placebo), and nightmares or abnormal dreams (0.9% at 5 mg and 2.2% at 10 mg vs. 0.9% for placebo).[3] Less common side effects include sleep paralysis (1.3% at 5 mg and 1.6% at 10 mg vs. 0% for placebo) and hypnagogic hallucinations (0.1% at 5 mg and 0.7% at 10 mg vs. 0% for placebo).[3]
Lemborexant at doses of 10, 20, and 30 mg produces drug-liking responses similar to those of zolpidem (30 mg) and suvorexant (40 mg) in recreational sedative drug users.[3] It is a controlled substance in the United States and is considered to have a low misuse potential.[3][17]
Pharmacology
Pharmacodynamics
Lemborexant is a dual antagonist of the orexin OX1 and OX2 receptors.[18][19][20] It associates and dissociates from the orexin receptors more rapidly than certain other orexin receptor antagonists, such as suvorexant, and this may cause it to have a shorter duration of action.[12]
Pharmacokinetics
The bioavailability of lemborexant is good and is at least 87%.[5][6] The time to peak levels of lemborexant is 1 to 3 hours.[3] A high-fat and high-calorie meal has been found to delay the time to peak levels by 2 hours.[3] Its plasma protein binding in vitro is 94%.[3] Lemborexant is metabolized primarily by CYP3A4 and to a lesser extent by CYP3A5.[3] The "effective" half-life of lemborexant is 17 to 19 hours while its terminal elimination half-life is 55 hours.[3][7][8] The medication is excreted in feces (57%) and to a lesser extent urine (29%).[3]
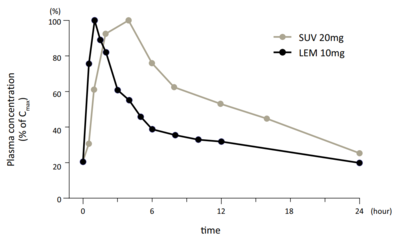
Although lemborexant has a longer terminal elimination half-life than suvorexant, it appears to be more rapidly cleared than suvorexant in the earlier phases of elimination.[21][7] In addition, lemborexant dissociates from the orexin receptors more rapidly than does suvorexant.[21] These differences may allow for comparatively reduced next-day effects such as daytime somnolence with lemborexant.[21][7]
History
In June 2016, Eisai initiated Phase III clinical trials in the United States, France, Germany, Italy, Japan, Poland, Spain and the UK.[22]
In December 2019, lemborexant was approved for use in the United States based on results from the SUNRISE 1 and SUNRISE 2 Phase III clinical trials.[11][23]
Society and culture
Names
Lemborexant is the generic name of the drug and its INN while E-2006 was its developmental code name. Lemborexant is sold under the brand name Dayvigo.[3]
Availability
Lemborexant is marketed in the United States, Canada, Australia, and Japan.[24][25][26][27] It is not approved by the European Medicines Agency (EMA) for use in the European Union or by the Medicines and Healthcare products Regulatory Agency (MHRA) in the United Kingdom.[28][29]
Research
Lemborexant is under development for the treatment of circadian rhythm sleep disorders, sleep apnea, and chronic obstructive pulmonary disease.[30] As of February 2022, it is in phase 2 clinical trials for circadian rhythm sleep disorders and phase 1 trials for sleep apnea and chronic obstructive pulmonary disease.[30]
References
- ^ a b "Dayvigo". Therapeutic Goods Administration (TGA). 23 July 2021. Retrieved 5 September 2021.
- ^ "Updates to the Prescribing Medicines in Pregnancy database". Therapeutic Goods Administration (TGA). 12 May 2022. Retrieved 13 May 2022.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad "Dayvigo- lemborexant tablet, film coated". DailyMed. Retrieved 17 June 2021.
- ^ "Summary Basis of Decision (SBD) for Dayvigo". Health Canada. 23 October 2014. Retrieved 29 May 2022.
- ^ a b Hoyer D, Jacobson LH (2018). "Lemborexant. Dual orexin receptor antagonist, Treatment of insomnia". Drugs of the Future. 43 (10): 715. doi:10.1358/dof.2018.043.10.2828699. ISSN 0377-8282.
- ^ a b Lalovic B, Majid O, Aluri J, Landry I, Moline M, Hussein Z (December 2020). "Population Pharmacokinetics and Exposure-Response Analyses for the Most Frequent Adverse Events Following Treatment With Lemborexant, an Orexin Receptor Antagonist, in Subjects With Insomnia Disorder". Journal of Clinical Pharmacology. 60 (12): 1642–1654. doi:10.1002/jcph.1683. PMC 7689791. PMID 32666570.
- ^ a b c d Muehlan C, Vaillant C, Zenklusen I, Kraehenbuehl S, Dingemanse J (November 2020). "Clinical pharmacology, efficacy, and safety of orexin receptor antagonists for the treatment of insomnia disorders". Expert Opinion on Drug Metabolism & Toxicology. 16 (11): 1063–1078. doi:10.1080/17425255.2020.1817380. PMID 32901578. S2CID 221572078.
- ^ a b c d e f g h i j Waters K (February 2022). "Review of the Efficacy and Safety of Lemborexant, a Dual Receptor Orexin Antagonist (DORA), in the Treatment of Adults With Insomnia Disorder". The Annals of Pharmacotherapy. 56 (2): 213–221. doi:10.1177/10600280211008492. PMID 34078141. S2CID 235321467.
- ^ "Novel Drug Approvals for 2019". U.S. Food and Drug Administration (FDA). 2 January 2020. Retrieved 10 January 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "FDA-Approved Drugs: Lemborexant". U.S. Food and Drug Administration (FDA). Retrieved 10 January 2020.
- ^ a b "FDA Approves Dayvigo (lemborexant) for the Treatment of Insomnia in Adult Patients". Drugs.com. 23 December 2019. Retrieved 10 January 2020.
- ^ a b Jacobson LH, Hoyer D, de Lecea L (May 2022). "Hypocretins (orexins): The ultimate translational neuropeptides". Journal of Internal Medicine. 291 (5): 533–556. doi:10.1111/joim.13406. PMID 35043499. S2CID 248119793.
- ^ Markham A (April 2022). "Daridorexant: First Approval". Drugs. 82 (5): 601–607. doi:10.1007/s40265-022-01699-y. PMC 9042981. PMID 35298826.
- ^ a b c De Crescenzo F, D'Alò GL, Ostinelli EG, Ciabattini M, Di Franco V, Watanabe N, et al. (July 2022). "Comparative effects of pharmacological interventions for the acute and long-term management of insomnia disorder in adults: a systematic review and network meta-analysis". Lancet. 400 (10347): 170–184. doi:10.1016/S0140-6736(22)00878-9. hdl:11380/1288245. PMID 35843245. S2CID 250536370.
- ^ Suzuki H, Hibino H (18 August 2021). "The effect of lemborexant for insomnia disorder". SAGE Open Medicine. 9: 20503121211039098. doi:10.1177/20503121211039098. PMC 8377315. PMID 34422270.
- ^ Murphy P, Kumar D, Zammit G, Rosenberg R, Moline M (May 2020). "Safety of lemborexant versus placebo and zolpidem: effects on auditory awakening threshold, postural stability, and cognitive performance in healthy older participants in the middle of the night and upon morning awakening". Journal of Clinical Sleep Medicine. 16 (5): 765–773. doi:10.5664/jcsm.8294. PMC 7849806. PMID 32022664.
- ^ Asakura S, Shiotani M, Gauvin DV, Fujiwara A, Ueno T, Bower N, et al. (December 2021). "Nonclinical evaluation of abuse liability of the dual orexin receptor antagonist lemborexant". Regulatory Toxicology and Pharmacology. 127: 105053. doi:10.1016/j.yrtph.2021.105053. PMID 34619288. S2CID 238476630.
- ^ Christopher JA (2014). "Small-molecule antagonists of the orexin receptors". Pharmaceutical Patent Analyst. 3 (6): 625–638. doi:10.4155/ppa.14.46. PMID 25489915.
- ^ Boss C, Roch C (August 2015). "Recent trends in orexin research--2010 to 2015". Bioorganic & Medicinal Chemistry Letters. 25 (15): 2875–2887. doi:10.1016/j.bmcl.2015.05.012. PMID 26045032.
- ^ Boss C (December 2014). "Orexin receptor antagonists--a patent review (2010 to August 2014)". Expert Opinion on Therapeutic Patents. 24 (12): 1367–1381. doi:10.1517/13543776.2014.978859. PMID 25407283. S2CID 21106711.
- ^ a b c d Kishi T, Nishida M, Koebis M, Taninaga T, Muramoto K, Kubota N, et al. (December 2021). "Evidence-based insomnia treatment strategy using novel orexin antagonists: A review". Neuropsychopharmacology Reports. 41 (4): 450–458. doi:10.1002/npr2.12205. PMC 8698673. PMID 34553844.
- ^ "Lemborexant". Specialist Pharmacy Service. Archived from the original on 7 November 2017. Retrieved 5 November 2017.
- ^ "Drug Trials Snapshot: Dayvigo". U.S. Food and Drug Administration (FDA). 20 December 2019. Retrieved 24 January 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Micromedex Products: Please Login".
- ^ "Drug Product Database: Access the database". 18 March 2010.
- ^ "Dayvigo". 23 July 2021.
- ^ "EISAI TO LAUNCH IN-HOUSE DEVELOPED NEW ANTI-INSOMNIA DRUG DAYVIGO® (LEMBOREXANT) WITH INDICATION FOR INSOMNIA IN JAPAN | News Release:2020 | Eisai Co., Ltd".
- ^ "Medicines". European Medicines Agency.
- ^ "Products". Medicines and Healthcare products Regulatory Agency (MHRA).
- ^ a b "Lemborexant - Eisai - AdisInsight".
