Glycocalyx
| Glycocalyx | |
|---|---|
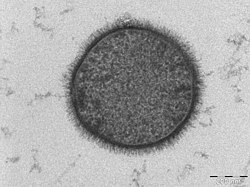 TEM micrograph of a B. subtilis bacterium, with the hair-like glycocalyx visible surrounding the cell membrane (scale bar = 200 nm) | |
| Identifiers | |
| MeSH | D019276 |
| TH | H1.00.01.1.00002 |
| FMA | 66838 |
| Anatomical terms of microanatomy | |
The glycocalyx (pl.: glycocalyces or glycocalyxes), also known as the pericellular matrix and cell coat, is a layer of glycoproteins and glycolipids which surround the cell membranes of bacteria, epithelial cells, and other cells. It was described in a review article in 1970.[1]
Animal epithelial cells have a fuzz-like coating on the external surface of their plasma membranes. This viscous coating is the glycocalyx that consists of several carbohydrate moieties of membrane glycolipids and glycoproteins, which serve as backbone molecules for support. Generally, the carbohydrate portion of the glycolipids found on the surface of plasma membranes helps these molecules contribute to cell–cell recognition, communication, and intercellular adhesion.[2]
The glycocalyx is a type of identifier that the body uses to distinguish between its own healthy cells and transplanted tissues, diseased cells, or invading organisms. Included in the glycocalyx are cell-adhesion molecules that enable cells to adhere to each other and guide the movement of cells during embryonic development.[3] The glycocalyx plays a major role in regulation of endothelial vascular tissue, including the modulation of red blood cell volume in capillaries.[4]
The term was initially applied to the polysaccharide matrix coating epithelial cells, but its functions have been discovered to go well beyond that.
In vascular endothelial tissue
The glycocalyx is located on the apical surface of vascular endothelial cells which line the lumen. When vessels are stained with cationic dyes such as Alcian blue stain, transmission electron microscopy shows a small, irregularly shaped layer extending approximately 50–100 nm into the lumen of a blood vessel. Another study used osmium tetroxide staining during freeze substitution, and showed that the endothelial glycocalyx could be up to 11 μm thick.[5] It is present throughout a diverse range of microvascular beds (capillaries) and macrovessels (arteries and veins). The glycocalyx also consists of a wide range of enzymes and proteins that regulate leukocyte and thrombocyte adherence, since its principal role in the vasculature is to maintain plasma and vessel-wall homeostasis. These enzymes and proteins include:
- Endothelial nitric oxide synthase (endothelial NOS)
- Extracellular superoxide dismutase (SOD3)
- Angiotensin converting enzyme
- Antithrombin-III
- Lipoprotein lipase
- Apolipoproteins
- Growth factors
- Chemokines
The enzymes and proteins listed above serve to reinforce the glycocalyx barrier against vascular and other diseases. Another main function of the glycocalyx within the vascular endothelium is that it shields the vascular walls from direct exposure to blood flow, while serving as a vascular permeability barrier.[6] Its protective functions are universal throughout the vascular system, but its relative importance varies depending on its exact location in the vasculature. In microvascular tissue, the glycocalyx serves as a vascular permeability barrier by inhibiting coagulation and leukocyte adhesion. Leukocytes must not stick to the vascular wall because they are important components of the immune system that must be able to travel to a specific region of the body when needed. In arterial vascular tissue, the glycocalyx also inhibits coagulation and leukocyte adhesion, but through mediation of shear stress-induced nitric oxide release. Another protective function throughout the cardiovascular system is its ability to affect the filtration of interstitial fluid from capillaries into the interstitial space.[7]
The glycocalyx, which is located on the apical surface of endothelial cells, is composed of a negatively charged network of proteoglycans, glycoproteins, and glycolipids.[8] Along the luminal surface of the vascular glycocalyx exists an empty layer that excludes red blood cells.[9]
Disruption and disease
Because the glycocalyx is so prominent throughout the cardiovascular system, disruption to this structure has detrimental effects that can cause disease. Certain stimuli that cause atheroma may lead to enhanced sensitivity of vasculature. Initial dysfunction of the glycocalyx can be caused by hyperglycemia or oxidized low-density lipoproteins (LDLs), which then causes atherothrombosis. In microvasculature, dysfunction of the glycocalyx leads to internal fluid imbalance, and potentially edema. In arterial vascular tissue, glycocalyx disruption causes inflammation and atherothrombosis.[10]
Experiments have been performed to test precisely how the glycocalyx can be altered or damaged. One particular study used an isolated perfused heart model designed to facilitate detection of the state of the vascular barrier portion, and sought to cause insult-induced shedding of the glycocalyx to ascertain the cause-and-effect relationship between glycocalyx shedding and vascular permeability. Hypoxic perfusion of the glycocalyx was thought to be sufficient to initiate a degradation mechanism of the endothelial barrier. The study found that flow of oxygen throughout the blood vessels did not have to be completely absent (ischemic hypoxia), but that minimal[clarification needed] levels of oxygen were sufficient to cause the degradation. Shedding of the glycocalyx can be triggered by inflammatory stimuli, such as tumor necrosis factor-alpha. Whatever the stimulus is, however, shedding of the glycocalyx leads to a drastic[clarification needed] increase in vascular permeability. Vascular walls being permeable is disadvantageous, since that would enable passage of some macromolecules or other harmful antigens.[11]
Other sources of damage to the endothelial glycocalyx have been observed in several pathological conditions such as inflammation,[12] hyperglycemia,[13] ischemia-reperfusion,[14] viral infections[15] and sepsis.[16]
Some key components of the glycocalyx such as syndecans, heparan sulphate, chondroitin sulphate and hyaluronan can be shed of the endothelial layer by enzymes. Hyaluronidase, hepararanse/heparinase, matrix and membrane-type matrix metalloproteases, thrombin, plasmin and elastase are some examples of enzymes that can induce shedding of the glycocalyx and these sheddases can therefor contribute to degradation of the glycocalyx layer in several pathological conditions.[17] Research shows that plasma hyaluronidase activity is decreased in experimental as well as in clinical septic shock and is therefore not considered to be a sheddase in sepsis.[18] Concomitant, the endogenous plasma inhibition of hyaluronidase is increased and could serve as a protection against glycocalyx shedding.
Fluid shear stress is also a potential problem if the glycocalyx is degraded for any reason. This type of frictional stress is caused by the movement of viscous fluid (i.e. blood) along the lumen boundary. Another similar experiment was carried out to determine what kinds of stimuli cause fluid shear stress. The initial measurement was taken with intravital microscopy, which showed a slow-moving plasma layer, the glycocalyx, of 1 μm thick. Light dye damaged the glycocalyx minimally, but that small change increased capillary hematocrit. Thus, fluorescence light microscopy should not be used to study the glycocalyx because that particular method uses a dye. The glycocalyx can also be reduced in thickness when treated with oxidized LDL.[19] These stimuli, along with many other factors, can cause damage to the delicate glycocalyx. These studies are evidence that the glycocalyx plays a crucial role in cardiovascular system health.
In bacteria and nature

A glycocalyx, literally meaning "sugar coat" (glykys = sweet, kalyx = husk), is a network of polysaccharides that project from cellular surfaces of bacteria, which classifies it as a universal surface component of a bacterial cell, found just outside the bacterial cell wall. A distinct, gelatinous glycocalyx is called a capsule, whereas an irregular, diffuse layer is called a slime layer. This coat is extremely hydrated and stains with ruthenium red.
Bacteria growing in natural ecosystems, such as in soil, bovine intestines, or the human urinary tract, are surrounded by some sort of glycocalyx-enclosed microcolony.[20] It serves to protect the bacterium from harmful phagocytes by creating capsules or allowing the bacterium to attach itself to inert surfaces, such as teeth or rocks, via biofilms (e.g. Streptococcus pneumoniae attaches itself to either lung cells, prokaryotes, or other bacteria which can fuse their glycocalices to envelop the colony).
In the digestive tract
A glycocalyx can also be found on the apical portion of microvilli within the digestive tract, especially within the small intestine. It creates a meshwork 0.3 μm thick and consists of acidic mucopolysaccharides and glycoproteins that project from the apical plasma membrane of epithelial absorptive cells. It provides additional surface for adsorption and includes enzymes secreted by the absorptive cells that are essential for the final steps of digestion of proteins and sugars.
Other generalized functions
- Protection: Cushions the plasma membrane and protects it from chemical injury
- Immunity to infection: Enables the immune system to recognize and selectively attack foreign organisms
- Defense against cancer: Changes in the glycocalyx of cancerous cells enable the immune system to recognize and destroy them.
- Transplant compatibility: Forms the basis for compatibility of blood transfusions, tissue grafts, and organ transplants
- Cell adhesion: Binds cells together so that tissues do not fall apart
- Inflammation regulation: Glycocalyx coating on endothelial walls in blood vessels prevents leukocytes from rolling/binding in healthy states.[21]
- Fertilization: Enables sperm to recognize and bind to eggs[22]
- Embryonic development: Guides embryonic cells to their destinations in the body
See also
References
- ^ Martínez-Palomo, A. (1970). "The Surface Coats of Animal Cells". International Review of Cytology. 29: 29–75. doi:10.1016/S0074-7696(08)60032-7. ISBN 9780123643292.
- ^ McKinley, M. & V.D. O’Loughlin. Human Anatomy. McGraw-Hill, 2012. 3rd ed. p. 30-31.
- ^ Saladin, Kenneth. "Anatomy & Physiology: The unity of form and function." McGraw Hill. 5th Edition. 2010. p. 94-95
- ^ Reitsma, Sietze. "The endothelial glycocalyx: composition, functions, and visualization." European Journal of Physiology. 2007. Vol. 454. Num. 3. p. 345-359
- ^ Ebong, Eno; Macaluso FP; Spray DC; Tarbell JM (August 2011). "Imaging the Endothelial Glycocalyx In Vitro By Rapid Freezing/Freeze Substitution Transmission Electron Microscopy". Arteriosclerosis, Thrombosis, and Vascular Biology. 31 (8): 1908–1915. doi:10.1161/ATVBAHA.111.225268. PMC 3141106. PMID 21474821.
- ^ Van de Berg, Bernard M., Max Nieuwdorp, Erik S.G. Stroes, Hans Vink. "Glycocalyx and endothelial (dys) function: from mice to men." Pharmacological Reports, 2006, 57: 75-80.
- ^ Drake-Holland, Angela & Mark Noble. "The Important New Drug Target in Cardiovascular Medicine – the Vascular Glycocalyx." Cardiovascular & Hematological Disorders-Drug Targets, 2009, 9, p. 118-123
- ^ Van de Berg, Bernard M., Max Nieuwdorp, Erik S.G. Stroes, Hans Vink. Glycocalyx and endothelial (dys) function: from mice to men. Pharmacological Reports, 2006, 57: 75-80.
- ^ Reitsma, Sietze; Slaaf, Dick W.; Vink, Hans; van Zandvoort, Marc A. M. J.; oude Egbrink, Mirjam G. A. (June 2007). "The endothelial glycocalyx: composition, functions, and visualization". Pflügers Archiv: European Journal of Physiology. 454 (3): 345–359. doi:10.1007/s00424-007-0212-8. PMC 1915585. PMID 17256154.
- ^ Drake-Holland, Angela & Mark Noble. "The Important New Drug Target in Cardiovascular Medicine – the Vascular Glycocalyx." Cardiovascular & Hematological Disorders-Drug Targets, 2009, 9, p. 118–123
- ^ Annecke, T., et al. "Shedding of the coronary endothelial glycocalyx: effects of hypoxia/reoxygenation vs ischaemia/reperfusion." British Journal of Anaesthesia, 2011. 107 (5): 679–86
- ^ Henry, Charmaine B. S.; Duling, Brian R. (2000-12-01). "TNF-α increases entry of macromolecules into luminal endothelial cell glycocalyx". American Journal of Physiology. Heart and Circulatory Physiology. 279 (6): H2815–H2823. doi:10.1152/ajpheart.2000.279.6.H2815. ISSN 0363-6135. PMID 11087236. S2CID 86646327.
- ^ Zuurbier, Coert J.; Demirci, Cihan; Koeman, Anneke; Vink, Hans; Ince, Can (October 2005). "Short-term hyperglycemia increases endothelial glycocalyx permeability and acutely decreases lineal density of capillaries with flowing red blood cells". Journal of Applied Physiology. 99 (4): 1471–1476. doi:10.1152/japplphysiol.00436.2005. ISSN 8750-7587. PMID 16024521.
- ^ Mulivor, A. W.; Lipowsky, H. H. (May 2004). "Inflammation- and ischemia-induced shedding of venular glycocalyx". American Journal of Physiology. Heart and Circulatory Physiology. 286 (5): H1672–H1680. doi:10.1152/ajpheart.00832.2003. ISSN 0363-6135. PMID 14704229.
- ^ Becker, Bernhard F.; Jacob, Matthias; Leipert, Stephanie; Salmon, Andrew H. J.; Chappell, Daniel (September 2015). "Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases: Endothelial glycocalyx - emerging clinical impact". British Journal of Clinical Pharmacology. 80 (3): 389–402. doi:10.1111/bcp.12629. PMC 4574825. PMID 25778676.
- ^ Steppan, Jochen; Hofer, Stefan; Funke, Benjamin; Brenner, Thorsten; Henrich, Michael; Martin, Eike; Weitz, Jürgen; Hofmann, Ursula; Weigand, Markus A. (January 2011). "Sepsis and major abdominal surgery lead to flaking of the endothelial glycocalix". The Journal of Surgical Research. 165 (1): 136–141. doi:10.1016/j.jss.2009.04.034. ISSN 1095-8673. PMID 19560161.
- ^ Becker, Bernhard F.; Jacob, Matthias; Leipert, Stephanie; Salmon, Andrew H. J.; Chappell, Daniel (September 2015). "Degradation of the endothelial glycocalyx in clinical settings: searching for the sheddases". British Journal of Clinical Pharmacology. 80 (3): 389–402. doi:10.1111/bcp.12629. ISSN 1365-2125. PMC 4574825. PMID 25778676.
- ^ van der Heijden, Jaap; Kolliopoulos, Constantinos; Skorup, Paul; Sallisalmi, Marko; Heldin, Paraskevi; Hultström, Michael; Tenhunen, Jyrki (2021-10-11). "Plasma hyaluronan, hyaluronidase activity and endogenous hyaluronidase inhibition in sepsis: an experimental and clinical cohort study". Intensive Care Medicine Experimental. 9 (1): 53. doi:10.1186/s40635-021-00418-3. ISSN 2197-425X. PMC 8502523. PMID 34632531.
- ^ Gouverneur, Mirella. Dissertation. "Fluid shear stress directly stimulates synthesis of the endothelial glycocalyx : perturbations by hyperglycemia." 2006. University of Amsterdam. p. 115–153
- ^ Costerton & Irvin. Bacterial Glycocalyx in Nature and Disease. Annual Reviews Microbiology, 1981. Vol. 35: p. 299-324
- ^ Near-Wall {micro}-PIV Reveals a Hydrodynamically Relevant Endothelial Surface Layer in Venules In Vivo - Smith et al. 85 (1): 637 - Biophysical Journal Archived 2008-12-03 at the Wayback Machine
- ^ Schroter, Sabine; Osterhoff, Caroline; McArdle, Wendy; Ivell, Richard (1999). "The glycocalyx of the sperm surface". Human Reproduction Update. 5 (4): 302–313. doi:10.1093/humupd/5.4.302. PMID 10465522.
External links
- Smart carbohydrate chemistry as a means to understand glycocalyx biology Archived 2016-07-10 at the Wayback Machine – Video by the Lindhorst group at Beilstein TV
