Vitamin B12
 General skeletal formula of cobalamins | |
 | |
| Clinical data | |
|---|---|
| Other names | Vitamin B12, vitamin B-12, cobalamin |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605007 |
| License data |
|
| Routes of administration | By mouth, sublingual, intravenous (IV), intramuscular (IM), intranasal |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Readily absorbed in the distal half of the ileum. |
| Protein binding | Very high to specific transcobalamins plasma proteins. Binding of hydroxocobalamin is slightly higher than cyanocobalamin. |
| Metabolism | Liver |
| Elimination half-life | Approximately 6 days (400 days in the liver). |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C63H88CoN14O14P |
| Molar mass | 1355.388 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Vitamin B12, also known as cobalamin, is a water-soluble vitamin involved in metabolism.[2] It is one of eight B vitamins. It is required by animals, which use it as a cofactor in DNA synthesis, and in both fatty acid and amino acid metabolism.[3] It is important in the normal functioning of the nervous system via its role in the synthesis of myelin, and in the circulatory system in the maturation of red blood cells in the bone marrow.[2][4] Plants do not need cobalamin and carry out the reactions with enzymes that are not dependent on it.[5]
Vitamin B12 is the most chemically complex of all vitamins,[6] and for humans the only vitamin that must be sourced from animal-derived foods or supplements.[2][7] Only some archaea and bacteria can synthesize vitamin B12.[8] Vitamin B12 deficiency is a widespread condition that is particularly prevalent in populations with low consumption of animal foods. Such diets can be due to a variety of reasons, such as low socioeconomic status, ethical considerations, or lifestyle choices such as veganism.[9]
Foods containing vitamin B12 include meat, shellfish, liver, fish, poultry, eggs, and dairy products.[2] Many breakfast cereals are fortified with the vitamin.[2] Supplements and medications are available to treat and prevent vitamin B12 deficiency.[2] They are usually taken by mouth, but for the treatment of deficiency may also be given as an intramuscular injection.[2][6]
Vitamin B12 deficiencies have a greater effect on young children, pregnant and elderly people, and are more common in middle and lower-developed countries due to malnutrition.[10] The most common cause of vitamin B12 deficiency in developed countries is impaired absorption due to a loss of gastric intrinsic factor (IF) which must be bound to a food source of B12 for absorption to occur.[11] A second major cause is an age-related decline in stomach acid production (achlorhydria), because acid exposure frees protein-bound vitamin.[12] For the same reason, people on long-term antacid therapy, using proton-pump inhibitors,[13] H2 blockers or other antacids are at increased risk.[14]
The diets of vegetarians and vegans may not provide sufficient B12 unless a dietary supplement is taken.[2] A deficiency may be characterized by limb neuropathy or a blood disorder called pernicious anemia, a type of anemia in which red blood cells become abnormally large.[2] This can result in fatigue, decreased ability to think, lightheadedness, shortness of breath, frequent infections, poor appetite, numbness in the hands and feet, depression, memory loss, confusion, difficulty walking, blurred vision, irreversible nerve damage, and many others.[15] If left untreated in infants, deficiency may lead to neurological damage and anemia.[2] Folate levels in the individual may affect the course of pathological changes and symptomatology of vitamin B12 deficiency. Vitamin B12 deficiency in pregnant women is strongly associated with an increased risk of spontaneous abortion, congenital malformations such as neural tube defects, and problems with brain development growth in the unborn child.[10]
Vitamin B12 was discovered as a result of pernicious anemia, an autoimmune disorder in which the blood has a lower than normal number of red blood cells, due to a deficiency of vitamin B12.[5][16] The ability to absorb the vitamin declines with age, especially in people over 60.[17]
Definition
Vitamin B12 is a coordination complex of cobalt, which occupies the center of a corrin ligand and is further bound to a benzimidazole ligand and adenosyl group.[18] Several related species are known and these behave similarly, in particular, all function as vitamins. This collection of compounds is sometimes referred to as "cobalamins". These chemical compounds have a similar molecular structure, each of which shows vitamin activity in a vitamin-deficient biological system, they are referred to as vitamers. The vitamin activity is as a coenzyme, meaning that its presence is required for some enzyme-catalyzed reactions.[12][19]
- adenosylcobalamin
- cyanocobalamin, the adenosyl ligand in vitamin B12 is replaced by cyanide.
- hydroxocobalamin, the adenosyl ligand in vitamin B12 is replaced by hydroxide.
- methylcobalamin, the adenosyl ligand in vitamin B12 is replaced by methyl.
Cyanocobalamin is a manufactured form of B12. Bacterial fermentation creates AdoB12 and MeB12, which are converted to cyanocobalamin by the addition of potassium cyanide in the presence of sodium nitrite and heat. Once consumed, cyanocobalamin is converted to the biologically active AdoB12 and MeB12. [citation needed]The two bioactive forms of vitamin B
12 are methylcobalamin in cytosol and adenosylcobalamin in mitochondria.[citation needed]
Cyanocobalamin is the most common form used in dietary supplements and food fortification because cyanide stabilizes the molecule against degradation. Methylcobalamin is also offered as a dietary supplement.[12] There is no advantage to the use of adenosylcobalamin or methylcobalamin forms for the treatment of vitamin B12 deficiency.[20][21][4]
Hydroxocobalamin can be injected intramuscularly to treat vitamin B12 deficiency. It can also be injected intravenously for the purpose of treating cyanide poisoning, as the hydroxyl group is displaced by cyanide, creating a non-toxic cyanocobalamin that is excreted in urine.
"Pseudovitamin B12" refers to compounds that are corrinoids with a structure similar to the vitamin but without vitamin activity.[22] Pseudovitamin B12 is the majority corrinoid in spirulina, an algal health food sometimes erroneously claimed as having this vitamin activity.[23]
Deficiency
Vitamin B12 deficiency can potentially cause severe and irreversible damage, especially to the brain and nervous system.[6][24] Deficiency at levels only slightly lower than normal can cause a range of symptoms such as fatigue, feeling weak, lightheadedness, dizziness, breathlessness, headaches, mouth ulcers, upset stomach, decreased appetite, difficulty walking (staggering balance problems),[15][25] muscle weakness, depression, poor memory, poor reflexes, confusion, and pale skin, feeling abnormal sensations, among others, especially in people over age 60.[6][15][26] Vitamin B12 deficiency can also cause symptoms of mania and psychosis.[27][28] Among other problems, weakened immunity, reduced fertility and interruption of blood circulation in women may occur.[29]
The main type of vitamin B12 deficiency anemia is pernicious anemia,[30] characterized by a triad of symptoms:
- Anemia with bone marrow promegaloblastosis (megaloblastic anemia). This is due to the inhibition of DNA synthesis (specifically purines and thymidine).
- Gastrointestinal symptoms: alteration in bowel motility, such as mild diarrhea or constipation, and loss of bladder or bowel control.[31] These are thought to be due to defective DNA synthesis inhibiting replication in tissue sites with a high turnover of cells. This may also be due to the autoimmune attack on the parietal cells of the stomach in pernicious anemia. There is an association with gastric antral vascular ectasia (which can be referred to as watermelon stomach), and pernicious anemia.[32]
- Neurological symptoms: sensory or motor deficiencies (absent reflexes, diminished vibration or soft touch sensation) and subacute combined degeneration of the spinal cord.[33] Deficiency symptoms in children include developmental delay, regression, irritability, involuntary movements and hypotonia.[34]
Vitamin B12 deficiency is most commonly caused by malabsorption, but can also result from low intake, immune gastritis, low presence of binding proteins, or use of certain medications.[6] Vegans—people who choose to not consume any animal-sourced foods—are at risk because plant-sourced foods do not contain the vitamin in sufficient amounts to prevent vitamin deficiency.[35] Vegetarians—people who consume animal byproducts such as dairy products and eggs, but not the flesh of any animal—are also at risk. Vitamin B12 deficiency has been observed in between 40% and 80% of the vegetarian population who do not also take a vitamin B12 supplement or consume vitamin-fortified food.[36] In Hong Kong and India, vitamin B12 deficiency has been found in roughly 80% of the vegan population. As with vegetarians, vegans can avoid this by consuming a dietary supplement or eating B12 fortified food such as cereal, plant-based milks, and nutritional yeast as a regular part of their diet.[37] The elderly are at increased risk because they tend to produce less stomach acid as they age, a condition known as achlorhydria, thereby increasing their probability of B12 deficiency due to reduced absorption.[2]
Nitrous oxide overdose or overuse converts the active monovalent form of vitamin B12 to the inactive bivalent form.[38]
Pregnancy, lactation, and early childhood
The U.S. Recommended Dietary Allowance (RDA) for pregnancy is 2.6 micrograms per day (μg/d), for lactation 2.8 μg/d. Determination of these values was based on an RDA of 2.4 μg/d for non-pregnant women, plus what will be transferred to the fetus during pregnancy and what will be delivered in breast milk.[12][39]: 972 However, looking at the same scientific evidence, the European Food Safety Authority (EFSA) sets adequate intake (AI) at 4.5 μg/d for pregnancy and 5.0 μg/d for lactation.[40] Low maternal vitamin B12, defined as serum concentration less than 148 pmol/L, increases the risk of miscarriage, preterm birth and newborn low birth weight.[41][39] During pregnancy the placenta concentrates B12, so that newborn infants have a higher serum concentration than their mothers.[12] As it is recently absorbed vitamin content that more effectively reaches the placenta, the vitamin consumed by the mother-to-be is more important than that contained in her liver tissue.[12][42]
Women who consume little animal-sourced food, or who are vegetarian or vegan, are at higher risk of becoming vitamin depleted during pregnancy than those who consume more animal products. This depletion can lead to anemia, and also an increased risk that their breastfed infants become vitamin deficient.[42][39] Vitamin B12 is not one of the supplements recommended by the World Health Organization for healthy women who are pregnant,[10] however, vitamin B12 is often suggested during pregnancy in a multivitamin along with folic acid[43][44] especially for pregnant mothers who follow a vegetarian or vegan diet.[45]
Low vitamin concentrations in human milk occur in families with low socioeconomic status or low consumption of animal products.[39]: 971, 973 Only a few countries, primarily in Africa, have mandatory food fortification programs for either wheat flour or maize flour; India has a voluntary fortification program.[46] What the nursing mother consumes is more important than her liver tissue content, as it is recently absorbed vitamin that more effectively reaches breast milk.[39]: 973 Breast milk B12 decreases over months of nursing in both well-nourished and vitamin-deficient mothers.[39]: 973–974 Exclusive or near-exclusive breastfeeding beyond six months is a strong indicator of low serum vitamin status in nursing infants. This is especially true when the vitamin status is poor during the pregnancy and if the early-introduced foods fed to the still-breastfeeding infant are vegan.[39]: 974–975
The risk of deficiency persists if the post-weaning diet is low in animal products.[39]: 974–975 Signs of low vitamin levels in infants and young children can include anemia, poor physical growth, and neurodevelopmental delays.[39]: 975 Children diagnosed with low serum B12 can be treated with intramuscular injections, then transitioned to an oral dietary supplement.[39]: 976
Gastric bypass surgery
Various methods of gastric bypass or gastric restriction surgery are used to treat morbid obesity. Roux-en-Y gastric bypass surgery (RYGB) but not sleeve gastric bypass surgery or gastric banding, increases the risk of vitamin B12 deficiency and requires preventive post-operative treatment with either injected or high-dose oral supplementation.[47][48][49] For post-operative oral supplementation, 1000 μg/d may be needed to prevent vitamin deficiency.[49]
Diagnosis
According to one review: "At present, no 'gold standard' test exists for the diagnosis of vitamin B12 deficiency and as a consequence the diagnosis requires consideration of both the clinical state of the patient and the results of investigations."[50] The vitamin deficiency is typically suspected when a routine complete blood count shows anemia with an elevated mean corpuscular volume (MCV). In addition, on the peripheral blood smear, macrocytes and hypersegmented polymorphonuclear leukocytes may be seen. Diagnosis is supported based on vitamin B12 blood levels below 150–180 pmol/L (200–250 pg/mL) in adults.[51] However, serum values can be maintained while tissue B12 stores are becoming depleted. Therefore, serum B12 values above the cut-off point of deficiency do not necessarily confirm adequate B12 status.[2] For this reason, elevated serum homocysteine over 15 micromol/L and methylmalonic acid (MMA) over 0.271 micromol/L are considered better indicators of B12 deficiency, rather than relying only on the concentration of B12 in blood.[2] However, elevated MMA is not conclusive, as it is seen in people with B12 deficiency, but also in elderly people who have renal insufficiency,[28] and elevated homocysteine is not conclusive, as it is also seen in people with folate deficiency.[52] In addition, elevated methylmalonic acid levels may also be related to metabolic disorders such as methylmalonic acidemia.[53] If nervous system damage is present and blood testing is inconclusive, a lumbar puncture may be carried out to measure cerebrospinal fluid B12 levels.[54]
Serum haptocorrin binds 80-90% of circulating B12, rendering it unavailable for cellular delivery by transcobalamin II. This is conjectured to be a circulating storage function.[55] Several serious, even life-threatening diseases cause elevated serum haptocorrin, measured as abnormally high serum vitamin B12, while at the same time potentially manifesting as a symptomatic vitamin deficiency because of insufficient vitamin bound to transcobalamin II which transfers the vitamin to cells.[56]
Medical uses
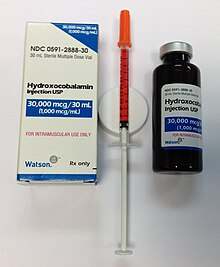
Treatment of deficiency
Severe vitamin B12 deficiency is initially corrected with daily intramuscular injections of 1000 μg of the vitamin, followed by maintenance via monthly injections of the same amount or daily oral dosing of 1000 μg. The oral daily dose far exceeds the vitamin requirement because the normal transporter protein-mediated absorption is absent, leaving only very inefficient intestinal passive absorption.[57][58] Injection side effects include skin rash, itching, chills, fever, hot flushes, nausea and dizziness. Oral maintenance treatment avoids this problem and significantly reduces the cost of treatment.[57][58]
Cyanide poisoning
For cyanide poisoning, a large amount of hydroxocobalamin may be given intravenously and sometimes in combination with sodium thiosulfate.[59][60] The mechanism of action is straightforward: the hydroxycobalamin hydroxide ligand is displaced by the toxic cyanide ion, and the resulting non-toxic cyanocobalamin is excreted in urine.[61]
Dietary recommendations
Some research shows that most people in the United States and the United Kingdom consume sufficient vitamin B12.[2][11] However, other research suggests that the proportion of people with low or marginal levels of vitamin B12 is up to 40% in the Western world.[2] Grain-based foods can be fortified by having the vitamin added to them. Vitamin B12 supplements are available as single or multivitamin tablets. Pharmaceutical preparations of vitamin B12 may be given by intramuscular injection.[6][62] Since there are few non-animal sources of the vitamin, vegans are advised to consume a dietary supplement or fortified foods for B12 intake, or risk serious health consequences.[6] Children in some regions of developing countries are at particular risk due to increased requirements during growth coupled with diets low in animal-sourced foods.
The US National Academy of Medicine updated estimated average requirements (EARs) and recommended dietary allowances (RDAs) for vitamin B12 in 1998.[6] The EAR for vitamin B12 for women and men ages 14 and up is 2.0 μg/day; the RDA is 2.4 μg/d. RDA is higher than EAR to identify amounts that will cover people with higher-than-average requirements. RDA for pregnancy equals 2.6 μg/day. RDA for lactation equals 2.8 μg/d. For infants up to 12 months, the adequate intake (AI) is 0.4–0.5 μg/day. (AIs are established when there is insufficient information to determine EARs and RDAs.) For children ages 1–13 years, the RDA increases with age from 0.9 to 1.8 μg/day. Because 10 to 30 percent of older people may be unable to effectively absorb vitamin B12 naturally occurring in foods, those older than 50 years should meet their RDA mainly by consuming foods fortified with vitamin B12 or a supplement containing vitamin B12. As for safety, tolerable upper intake levels (known as ULs) are set for vitamins and minerals when evidence is sufficient. In the case of vitamin B12 there is no UL, as there is no human data for adverse effects from high doses. Collectively the EARs, RDAs, AIs, and ULs are referred to as dietary reference intakes (DRIs).[12]
The European Food Safety Authority (EFSA) refers to the collective set of information as "dietary reference values", with population reference intake (PRI) instead of RDA, and average requirement instead of EAR. AI and UL are defined by EFSA the same as in the United States. For women and men over age 18, the adequate intake (AI) is set at 4.0 μg/day. AI for pregnancy is 4.5 μg/day, and for lactation 5.0 μg/day. For children aged 1–14 years, the AIs increase with age from 1.5 to 3.5 μg/day. These AIs are higher than the U.S. RDAs.[40] The EFSA also reviewed the safety question and reached the same conclusion as in the United States—that there was not sufficient evidence to set a UL for vitamin B12.[63]
The Japan National Institute of Health and Nutrition set the RDA for people ages 12 and older at 2.4 μg/day.[64] The World Health Organization also uses 2.4 μg/day as the adult recommended nutrient intake for this vitamin.[65]
For U.S. food and dietary supplement labeling purposes, the amount in a serving is expressed as a "percent of daily value" (%DV). For vitamin B12 labeling purposes, 100% of the daily value was 6.0 μg, but on 27 May 2016, it was revised downward to 2.4 μg (see Reference Daily Intake).[66][67] Compliance with the updated labeling regulations was required by 1 January 2020 for manufacturers with US$10 million or more in annual food sales, and by 1 January 2021 for manufacturers with lower volume food sales.[68][69]
Sources
Bacteria and archaea
Vitamin B12 is produced in nature by certain bacteria, and archaea.[70][71][72] It is synthesized by some bacteria in the gut microbiota in humans and other animals, but it has long been thought that humans cannot absorb this as it is made in the colon, downstream from the small intestine, where the absorption of most nutrients occurs.[73] Ruminants, such as cows and sheep, are foregut fermenters, meaning that plant food undergoes microbial fermentation in the rumen before entering the true stomach (abomasum), and thus they are absorbing vitamin B12 produced by bacteria.[73][74]
Other mammalian species (examples: rabbits, pikas, beaver, guinea pigs) consume high-fiber plants which pass through the gastrointestinal tract and undergo bacterial fermentation in the cecum and large intestine. In this hindgut fermentation, the material from the cecum is expelled as "cecotropes" and are re-ingested, a practice referred to as cecotrophy. Re-ingestion allows for absorption of nutrients made available by bacterial fermentation, and also of vitamins and other nutrients synthesized by the gut bacteria, including vitamin B12.[74]
Non-ruminant, non-hindgut herbivores may have an enlarged forestomach and/or small intestine to provide a place for bacterial fermentation and B-vitamin production, including B12.[74] For gut bacteria to produce vitamin B12, the animal must consume sufficient amounts of cobalt.[75] Soil that is deficient in cobalt may result in B12 deficiency, and B12 injections or cobalt supplementation may be required for livestock.[76]
Animal-derived foods
Animals store vitamin B12 from their diets in their livers and muscles and some pass the vitamin into their eggs and milk. Meat, liver, eggs, and milk are therefore sources of the vitamin for other animals, including humans.[62][2][77] For humans, the bioavailability from eggs is less than 9%, compared to 40% to 60% from fish, fowl, and meat.[78] Insects are a source of B12 for animals (including other insects and humans).[77][79] Animal-derived food sources with a high concentration of vitamin B12 include liver and other organ meats from lamb, veal, beef, and turkey; also shellfish and crab meat.[6][62][80]
Plants and algae
There is some evidence that bacterial fermentation of plant foods and symbiotic relationships between algae and bacteria can provide vitamin B12. However, the Academy of Nutrition and Dietetics considers plant and algae sources "unreliable", stating that vegans should turn to fortified foods and supplements instead.[35]
Natural plant and algae sources of vitamin B12 include fermented plant foods such as tempeh[81][82] and seaweed-derived foods such as nori and laverbread.[83][84][85] Methylcobalamin has been identified in Chlorella vulgaris.[86] Since only bacteria and some archea possess the genes and enzymes necessary to synthesize vitamin B12, plant and algae sources all obtain the vitamin secondarily from symbiosis with various species of bacteria,[5] or in the case of fermented plant foods, from bacterial fermentation.[81] Pseudovitamin B12 is the majority corrinoid in spirulina, an algal health food sometimes erroneously claimed as having vitamin activity.[23]
Fortified foods
Foods for which vitamin B12-fortified versions are available include breakfast cereals, plant-derived milk substitutes such as soy milk and oat milk, energy bars, and nutritional yeast.[80] The fortification ingredient is cyanocobalamin. Microbial fermentation yields adenosylcobalamin, which is then converted to cyanocobalamin by the addition of potassium cyanide or thiocyanate in the presence of sodium nitrite and heat.[87]
As of 2019, nineteen countries require food fortification of wheat flour, maize flour, or rice with vitamin B12. Most of these are in southeast Africa or Central America.[46]
Vegan advocacy organizations, among others, recommend that every vegan consume B12 from either fortified foods or supplements.[6][37][88][89]
Supplements

Vitamin B12 is included in multivitamin pills; in some countries grain-based foods such as bread and pasta are fortified with B12. In the US, non-prescription products can be purchased providing up to 5,000 μg each, and it is a common ingredient in energy drinks and energy shots, usually at many times the recommended dietary allowance of B12. The vitamin can also be supplied on prescription and delivered via injection or other means.[2]
A vitamer methylcobalamin is available as a supplement. The claimed advantage of such product is that methylcobalamin, unline cyanocobalamin, does not contain cyanide. The metabolic fate and biological distribution of methylcobalamin are expected to be similar to that of other sources of vitamin B12 in the diet.[90] The amount of cyanide in cyanocobalamin is generally not a concern, even in the 1,000 μg dose, since the amount of cyanide there (20 μg in a 1,000 μg cyanocobalamin tablet) is less than the daily consumption of cyanide from food, and therefore cyanocobalamin is not considered a health risk.[90] Besides that, the efficacy of methylcobalamin administration in treating vitamin B12 deficiency remains uncertain. While directly providing active cobalamin forms to deficient patients is an attractive approach promoted by the manufacturers of methylcobalamin products, it is not known whether methylcobalamin can reach its intracellular targets in its original, unmodified form to function effectively as ready coenzyme. It is also not known whether current level of evidence sufficient to recommend this relatively expensive strategy as an alternative to cyanocobalamin or hydroxocobalamin. There is currently insufficient evidence on comparative effectiveness and safety of various B12 vitamers (methylcobalamin, cyancobalamin, hydroxocobalamin, adenosylcobalamin).[91][92][93]
Intramuscular or intravenous injection
Injection of hydroxycobalamin is often used if digestive absorption is impaired,[2] but this course of action may not be necessary with high-dose oral supplements (such as 0.5–1.0 mg or more),[94][95] because with large quantities of the vitamin taken orally, even the 1% to 5% of free crystalline B12 that is absorbed along the entire intestine by passive diffusion may be sufficient to provide a necessary amount.[96]
A person with cobalamin C disease, a rare autosomal, recessive, inheritance disease which results in combined methylmalonic aciduria and homocystinuria),[97] can be treated with intravenous or intramuscular hydroxocobalamin.[98]
Nanotechnologies used in vitamin B12 supplementation
Conventional administration does not ensure specific distribution and controlled release of vitamin B12. Moreover, therapeutic protocols involving injection require health care people and commuting of patients to the hospital thus increasing the cost of the treatment and impairing the lifestyle of patients. Targeted delivery of vitamin B12 is a major focus of modern prescriptions. For example, conveying the vitamin to the bone marrow and nerve cells would help myelin recovery. Currently, several nanocarriers strategies are being developed for improving vitamin B12 delivery to simplify administration, reduce costs, improve pharmacokinetics, and ameliorate the quality of patients' lives.[99]
Pseudovitamin-B12
Pseudovitamin-B12 refers to B12-like analogues that are biologically inactive in humans.[22] Most cyanobacteria, including Spirulina, and some algae, such as Porphyra tenera (used to make a dried seaweed food called nori in Japan), have been found to contain mostly pseudovitamin-B12 instead of biologically active B12.[23][100] These pseudo-vitamin compounds can be found in some types of shellfish,[22] in edible insects,[101] and at times as metabolic breakdown products of cyanocobalamin added to dietary supplements and fortified foods.[102]
Pseudovitamin-B12 can show up as biologically active vitamin B12 when a microbiological assay with Lactobacillus delbrueckii subsp. lactis is used, as the bacteria can utilize the pseudovitamin despite it being unavailable to humans. To get a reliable reading of B12 content, more advanced techniques are available. One such technique involves pre-separation by silica gel and then assessment with B12-dependent E. coli bacteria.[22]
A related concept is antivitamin B12, compounds (often synthetic B12 analogues) that not only have no vitamin action but also actively interfere with the activity of true vitamin B12. The design of these compounds mainly involves the replacement of the metal ion with rhodium, nickel, or zinc; or the attachment of an inactive ligand such as 4-ethylphenyl. These compounds have the potential to be used for analyzing B12 utilization pathways or even attacking B12-dependent pathogens.[103]
Drug interactions
H2-receptor antagonists and proton-pump inhibitors
Gastric acid is needed to release vitamin B12 from protein for absorption. Reduced secretion of gastric acid and pepsin, from the use of H2 blocker or proton-pump inhibitor (PPI) drugs, can reduce the absorption of protein-bound (dietary) vitamin B12, although not of supplemental vitamin B12. H2-receptor antagonist examples include cimetidine, famotidine, nizatidine, and ranitidine. PPIs examples include omeprazole, lansoprazole, rabeprazole, pantoprazole, and esomeprazole. Clinically significant vitamin B12 deficiency and megaloblastic anemia are unlikely, unless these drug therapies are prolonged for two or more years, or if in addition, the person's dietary intake is below recommended levels. Symptomatic vitamin deficiency is more likely if the person is rendered achlorhydric (a complete absence of gastric acid secretion), which occurs more frequently with proton pump inhibitors than H2 blockers.[104]
Metformin
Reduced serum levels of vitamin B12 occur in up to 30% of people taking long-term anti-diabetic metformin.[105][106] Deficiency does not develop if dietary intake of vitamin B12 is adequate or prophylactic B12 supplementation is given. If the deficiency is detected, metformin can be continued while the deficiency is corrected with B12 supplements.[107]
Other drugs
Certain medications can decrease the absorption of orally consumed vitamin B12, including colchicine, extended-release potassium products, and antibiotics such as gentamicin, neomycin and tobramycin.[108] Anti-seizure medications phenobarbital, pregabalin, primidone and topiramate are associated with lower than normal serum vitamin concentration. However, serum levels were higher in people prescribed valproate.[109] In addition, certain drugs may interfere with laboratory tests for the vitamin, such as amoxicillin, erythromycin, methotrexate and pyrimethamine.[108]
Chemistry

Vitamin B12 is the most chemically complex of all the vitamins.[6] The structure of B12 is based on a corrin ring, which is similar to the porphyrin ring found in heme. The central metal ion is cobalt. As isolated as an air-stable solid and available commercially, cobalt in vitamin B12 (cyanocobalamin and other vitamers) is present in its +3 oxidation state. Biochemically, the cobalt center can take part in both two-electron and one-electron reductive processes to access the "reduced" (B12r, +2 oxidation state) and "super-reduced" (B12s, +1 oxidation state) forms. The ability to shuttle between the +1, +2, and +3 oxidation states is responsible for the versatile chemistry of vitamin B12, allowing it to serve as a donor of deoxyadenosyl radical (radical alkyl source) and as a methyl cation equivalent (electrophilic alkyl source).[110]

Four of the six coordination sites are provided by the corrin ring and a fifth by a dimethylbenzimidazole group. The sixth coordination site, the reactive center, is variable, being a cyano group (–CN), a hydroxyl group (–OH), a methyl group (–CH3) or a 5′-deoxyadenosyl group. Historically, the covalent carbon–cobalt bond is one of the first examples of carbon-metal bonds to be discovered in biology. The hydrogenases and, by necessity, enzymes associated with cobalt utilization, involve metal-carbon bonds.[111] Animals can convert cyanocobalamin and hydroxocobalamin to the bioactive forms adenosylcobalamin and methylcobalamin by enzymatically replacing the cyano or hydroxyl groups.
Methods for the analysis of vitamin B12 in food
Several methods have been used to determine the vitamin B12 content in foods including microbiological assays, chemiluminescence assays, polarographic, spectrophotometric, and high-performance liquid chromatography processes.[112] The microbiological assay has been the most commonly used assay technique for foods, utilizing certain vitamin B12-requiring microorganisms, such as Lactobacillus delbrueckii subsp. lactis ATCC7830.[78] However, it is no longer the reference method due to the high measurement uncertainty of vitamin B12.[113]
Furthermore, this assay requires overnight incubation and may give false results if any inactive vitamin B12 analogues are present in the foods.[114] Currently, radioisotope dilution assay (RIDA) with labeled vitamin B12 and hog IF (pigs) have been used to determine vitamin B12 content in food.[78] Previous reports have suggested that the RIDA method can detect higher concentrations of vitamin B12 in foods compared to the microbiological assay method.[78][112]
Biochemistry
Coenzyme function
Vitamin B12 functions as a coenzyme, meaning that its presence is required in some enzyme-catalyzed reactions.[12][19] Listed here are the three classes of enzymes that sometimes require B12 to function (in animals):
- Isomerases
- Rearrangements in which a hydrogen atom is directly transferred between two adjacent atoms with concomitant exchange of the second substituent, X, which may be a carbon atom with substituents, an oxygen atom of an alcohol, or an amine. These use the AdoB12 (adenosylcobalamin) form of the vitamin.[115]
- Methyltransferases
- Methyl (–CH3) group transfers between two molecules. These use the MeB12 (methylcobalamin) form of the vitamin.[116]
- Dehalogenases
In humans, two major coenzyme B12-dependent enzyme families corresponding to the first two reaction types, are known. These are typified by the following two enzymes:
Methylmalonyl-CoA mutase

Methylmalonyl coenzyme A mutase (MUT) is an isomerase enzyme that uses the AdoB12 form and reaction type 1 to convert L-methylmalonyl-CoA to succinyl-CoA, an important step in the catabolic breakdown of some amino acids into succinyl-CoA, which then enters energy production via the citric acid cycle.[115] This functionality is lost in vitamin B12 deficiency, and can be measured clinically as an increased serum methylmalonic acid (MMA) concentration. The MUT function is necessary for proper myelin synthesis.[4] Based on animal research, it is thought that the increased methylmalonyl-CoA hydrolyzes to form methylmalonate (methylmalonic acid), a neurotoxic dicarboxylic acid, causing neurological deterioration.[119]
Methionine synthase

Methionine synthase, coded by MTR gene, is a methyltransferase enzyme which uses the MeB12 and reaction type 2 to transfer a methyl group from 5-methyltetrahydrofolate to homocysteine, thereby generating tetrahydrofolate (THF) and methionine.[116] This functionality is lost in vitamin B12 deficiency, resulting in an increased homocysteine level and the trapping of folate as 5-methyl-tetrahydrofolate, from which THF (the active form of folate) cannot be recovered. THF plays an important role in DNA synthesis, so reduced availability of THF results in ineffective production of cells with rapid turnover, in particular red blood cells, and also intestinal wall cells which are responsible for absorption. THF may be regenerated via MTR or may be obtained from fresh folate in the diet. Thus all of the DNA synthetic effects of B12 deficiency, including the megaloblastic anemia of pernicious anemia, resolve if sufficient dietary folate is present. Thus the best-known "function" of B12 (that which is involved with DNA synthesis, cell division, and anemia) is a facultative function that is mediated by B12-conservation of an active form of folate which is needed for efficient DNA production.[116] Other cobalamin-requiring methyltransferase enzymes are also known in bacteria, such as Me-H4-MPT, coenzyme M methyltransferase.[120]
Physiology
Absorption
Vitamin B12 is absorbed by a B12-specific transport proteins or via passive diffusion.[12] Transport-mediated absorption and tissue delivery is a complex process involving three transport proteins: haptocorrin (HC), intrinsic factor (IF) and transcobalamin II (TC2), and respective membrane receptor proteins. HC is present in saliva. As vitamin-containing food is digested by hydrochloric acid and pepsin secreted into the stomach, HC binds the vitamin and protects it from acidic degradation.[12][121] Upon leaving the stomach the hydrochloric acid of the chyme is neutralized in the duodenum by bicarbonate,[122] and pancreatic proteases release the vitamin from HC, making it available to be bound by IF, which is a protein secreted by gastric parietal cells in response to the presence of food in the stomach. IF delivers the vitamin to receptor proteins cubilin and amnionless, which together form the cubam receptor in the distal ileum. The receptor is specific to the IF-B12 complex, and so will not bind to any vitamin content that is not bound to IF.[12][121]
Investigations into the intestinal absorption of B12 confirm that the upper limit of absorption per single oral dose is about 1.5 μg, with 50% efficiency. In contrast, the passive diffusion process of B12 absorption — normally a very small portion of total absorption of the vitamin from food consumption — may exceed the haptocorrin- and IF-mediated absorption when oral doses of B12 are very large, with roughly 1% efficiency. Thus, dietary supplement B12 supplementation at 500 to 1000 μg per day allows pernicious anemia and certain other defects in B12 absorption to be treated with daily oral megadoses of B12 without any correction of the underlying absorption defects.[121]
After the IF/B12 complex binds to cubam the complex is disassociated and the free vitamin is transported into the portal circulation. The vitamin is then transferred to TC2, which serves as the circulating plasma transporter, hereditary defects in the production of TC2 and its receptor may produce functional deficiencies in B12 and infantile megaloblastic anemia, and abnormal B12 related biochemistry, even in some cases with normal blood B12 levels. For the vitamin to serve inside cells, the TC2-B12 complex must bind to a cell receptor protein and be endocytosed. TC2 is degraded within a lysosome, and free B12 is released into the cytoplasm, where it is transformed into the bioactive coenzyme by cellular enzymes.[121][123]
Malabsorption
Antacid drugs that neutralize stomach acid and drugs that block acid production (such as proton-pump inhibitors) will inhibit the absorption of B12 by preventing the release from food in the stomach.[124] Other causes of B12 malabsorption include intrinsic factor deficiency, pernicious anemia, bariatric surgery pancreatic insufficiency, obstructive jaundice, tropical sprue and celiac disease, and radiation enteritis of the distal ileum.[121] Age can be a factor. Elderly people are often achlorhydric due to reduced stomach parietal cell function, and thus have an increased risk of B12 deficiency.[125]
Storage and excretion
How fast B12 levels change depends on the balance between how much B12 is obtained from the diet, how much is secreted and how much is absorbed. The total amount of vitamin B12 stored in the body is about 2–5 mg in adults. Around 50% of this is stored in the liver. Approximately 0.1% of this is lost per day by secretions into the gut, as not all these secretions are reabsorbed. Bile is the main form of B12 excretion; most of the B12 secreted in the bile is recycled via enterohepatic circulation. Excess B12 beyond the blood's binding capacity is typically excreted in urine. Owing to the extremely efficient enterohepatic circulation of B12, the liver can store 3 to 5 years' worth of vitamin B12; therefore, nutritional deficiency of this vitamin is rare in adults in the absence of malabsorption disorders.[12] In the absence of intrinsic factor or distal ileum receptors, only months to a year of vitamin B12 are stored.[126]
Cellular reprogramming
Vitamin B12 through its involvement in one-carbon metabolism plays a key role in cellular reprogramming and tissue regeneration and epigenetic regulation. Cellular reprogramming is the process by which somatic cells can be converted to a pluripotent state. Vitamin B12 levels affect the histone modification H3K36me3, which suppresses illegitimate transcription outside of gene promoters. Mice undergoing in vivo reprogramming were found to become depleted in B12 and show signs of methionine starvation while supplementing reprogramming mice and cells with B12 increased reprogramming efficiency, indicating a cell-intrinsic effect.[127][128]
Synthesis
Biosynthesis
Vitamin B12 is derived from a tetrapyrrolic structural framework created by the enzymes deaminase and cosynthetase which transform aminolevulinic acid via porphobilinogen and hydroxymethylbilane to uroporphyrinogen III. The latter is the first macrocyclic intermediate common to heme, chlorophyll, siroheme and B12 itself.[129][130] Later steps, especially the incorporation of the additional methyl groups of its structure, were investigated using 13C methyl-labelled S-adenosyl methionine. It was not until a genetically engineered strain of Pseudomonas denitrificans was used, in which eight of the genes involved in the biosynthesis of the vitamin had been overexpressed, that the complete sequence of methylation and other steps could be determined, thus fully establishing all the intermediates in the pathway.[131][132]
Species from the following genera and the following individual species are known to synthesize B12: Propionibacterium shermanii, Pseudomonas denitrificans, Streptomyces griseus, Acetobacterium, Aerobacter, Agrobacterium, Alcaligenes, Azotobacter, Bacillus, Clostridium, Corynebacterium, Flavobacterium, Lactobacillus, Micromonospora, Mycobacterium, Nocardia, Proteus, Rhizobium, Salmonella, Serratia, Streptococcus and Xanthomonas.[133][134]
Industrial
Industrial production of B12 is achieved through fermentation of selected microorganisms.[135] Streptomyces griseus, a bacterium once thought to be a fungus, was the commercial source of vitamin B12 for many years.[136] The species Pseudomonas denitrificans and Propionibacterium freudenreichii subsp. shermanii are more commonly used today.[135] These are grown under special conditions to enhance yield. Rhone-Poulenc improved yield via genetic engineering P. denitrificans.[137] Propionibacterium, the other commonly used bacteria, produce no exotoxins or endotoxins and are generally recognized as safe (have been granted GRAS status) by the Food and Drug Administration of the United States.[138]
The total world production of vitamin B12 in 2008 was 35,000 kg (77,175 lb).[139]
Laboratory
The complete laboratory synthesis of B12 was achieved by Robert Burns Woodward[140] and Albert Eschenmoser in 1972.[141][142] The work required the effort of 91 postdoctoral fellows (mostly at Harvard) and 12 PhD students (at ETH Zurich) from 19 nations. The synthesis constitutes a formal total synthesis, since the research groups only prepared the known intermediate cobyric acid, whose chemical conversion to vitamin B12 was previously reported. This synthesis of vitamin B12 is of no practical consequence due to its length, taking 72 chemical steps and giving an overall chemical yield well under 0.01%.[143] Although there have been sporadic synthetic efforts since 1972,[142] the Eschenmoser–Woodward synthesis remains the only completed (formal) total synthesis.
History
Descriptions of deficiency effects
Between 1849 and 1887, Thomas Addison described a case of pernicious anemia, William Osler and William Gardner first described a case of neuropathy, Hayem described large red cells in the peripheral blood in this condition, which he called "giant blood corpuscles" (now called macrocytes), Paul Ehrlich identified megaloblasts in the bone marrow, and Ludwig Lichtheim described a case of myelopathy.[144]
Identification of liver as an anti-anemia food
During the 1920s, George Whipple discovered that ingesting large amounts of raw liver seemed to most rapidly cure the anemia of blood loss in dogs, and hypothesized that eating liver might treat pernicious anemia.[145] Edwin Cohn prepared a liver extract that was 50 to 100 times more potent in treating pernicious anemia than the natural liver products. William Castle demonstrated that gastric juice contained an "intrinsic factor" which when combined with meat ingestion resulted in absorption of the vitamin in this condition.[144] In 1934, George Whipple shared the 1934 Nobel Prize in Physiology or Medicine with William P. Murphy and George Minot for discovery of an effective treatment for pernicious anemia using liver concentrate, later found to contain a large amount of vitamin B12.[144][146]
Identification of the active compound
While working at the Bureau of Dairy Industry, U.S. Department of Agriculture, Mary Shaw Shorb was assigned work on the bacterial strain Lactobacillus lactis Dorner (LLD), which was used to make yogurt and other cultured dairy products. The culture medium for LLD required liver extract. Shorb knew that the same liver extract was used to treat pernicious anemia (her father-in-law had died from the disease), and concluded that LLD could be developed as an assay method to identify the active compound. While at the University of Maryland, she received a small grant from Merck, and in collaboration with Karl Folkers from that company, developed the LLD assay. This identified "LLD factor" as essential for the bacteria's growth.[147] Shorb, Folker and Alexander R. Todd, at the University of Cambridge, used the LLD assay to extract the anti-pernicious anemia factor from liver extracts, purify it, and name it vitamin B12.[148] In 1955, Todd helped elucidate the structure of the vitamin. The complete chemical structure of the molecule was determined by Dorothy Hodgkin based on crystallographic data and published in 1955[149] and 1956,[150] for which, and for other crystallographic analyses, she was awarded the Nobel Prize in Chemistry in 1964.[151] Hodgkin went on to decipher the structure of insulin.[151]
George Whipple, George Minot and William Murphy were awarded the Nobel Prize in 1934 for their work on the vitamin. Three other Nobel laureates, Alexander R. Todd (1957), Dorothy Hodgkin (1964) and Robert Burns Woodward (1965) made important contributions to its study.[152]
Commercial production
Industrial production of vitamin B12 is achieved through fermentation of selected microorganisms.[135] As noted above, the completely synthetic laboratory synthesis of B12 was achieved by Robert Burns Woodward and Albert Eschenmoser in 1972, though this process has no commercial potential, requiring more than 70 steps and having a yield well below 0.01%.[143]
Society and culture
In the 1970s, John A. Myers, a physician residing in Baltimore, developed a program of injecting vitamins and minerals intravenously for various medical conditions. The formula included 1000 μg of cyanocobalamin. This came to be known as the Myers' cocktail. After he died in 1984, other physicians and naturopaths took up prescribing "intravenous micronutrient therapy" with unsubstantiated health claims for treating fatigue, low energy, stress, anxiety, migraine, depression, immunocompromised, promoting weight loss, and more.[153] However, other than a report on case studies[153] there are no benefits confirmed in the scientific literature.[154] Healthcare practitioners at clinics and spas prescribe versions of these intravenous combination products, but also intramuscular injections of just vitamin B12. A Mayo Clinic review concluded that there is no solid evidence that vitamin B12 injections provide an energy boost or aid weight loss.[155]
There is evidence that for elderly people, physicians often repeatedly prescribe and administer cyanocobalamin injections inappropriately, evidenced by the majority of subjects in one large study either having had normal serum concentrations or having not been tested before the injections.[156]
See also
Further reading
- Gherasim C, Lofgren M, Banerjee R (May 2013). "Navigating the B(12) road: assimilation, delivery, and disorders of cobalamin". J. Biol. Chem. 288 (19): 13186–13193. doi:10.1074/jbc.R113.458810. PMC 3650358. PMID 23539619.
References
- ^ Prieto T, Neuburger M, Spingler B, Zelder F (2016). "Inorganic Cyanide as Protecting Group in the Stereospecific Reconstitution of Vitamin B12 from an Artificial Green Secocorrinoid" (PDF). Org. Lett. 18 (20): 5292–5295. doi:10.1021/acs.orglett.6b02611. PMID 27726382.
- ^ a b c d e f g h i j k l m n o p q r Office of Dietary Supplements (6 April 2021). "Vitamin B12: Fact Sheet for Health Professionals". Bethesda, Maryland: US National Institutes of Health. Archived from the original on 8 October 2021. Retrieved 24 December 2021.
- ^ Yamada K (2013). "Cobalt: Its Role in Health and Disease". In Sigel A, Sigel H, Sigel RK (eds.). Interrelations between Essential Metal Ions and Human Diseases. Metal Ions in Life Sciences. Vol. 13. Springer. pp. 295–320. doi:10.1007/978-94-007-7500-8_9. ISBN 978-94-007-7499-5. PMID 24470095.
- ^ a b c Calderón-Ospina CA, Nava-Mesa MO (January 2020). "B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin". CNS Neurosci Ther. 26 (1): 5–13. doi:10.1111/cns.13207. PMC 6930825. PMID 31490017.
- ^ a b c Smith AG (21 September 2019). "Plants need their vitamins too". Current Opinion in Plant Biology. 10 (3): 266–275. doi:10.1016/j.pbi.2007.04.009. PMID 17434786.
- ^ a b c d e f g h i j k "Vitamin B12". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis, OR. 4 June 2015. Archived from the original on 29 October 2019. Retrieved 5 April 2019.
- ^ Vincenti A, Bertuzzo L, Limitone A, D'Antona G, Cena H (June 2021). "Perspective: Practical Approach to Preventing Subclinical B12 Deficiency in Elderly Population". Nutrients. 13 (6): 1913. doi:10.3390/nu13061913. PMC 8226782. PMID 34199569.
- ^ Watanabe F, Bito T (January 2018). "Vitamin B12 sources and microbial interaction". Exp Biol Med (Maywood). 243 (2): 148–158. doi:10.1177/1535370217746612. PMC 5788147. PMID 29216732.
- ^ Obeid R, Heil SG, Verhoeven MM, van den Heuvel EG, de Groot LC, Eussen SJ (2019). "Vitamin B12 Intake From Animal Foods, Biomarkers, and Health Aspects". Front Nutr. 6: 93. doi:10.3389/fnut.2019.00093. PMC 6611390. PMID 31316992.
- ^ a b c Finkelstein JL, Fothergill A, Venkatramanan S, Layden AJ, Williams JL, Crider KS, et al. (Cochrane Pregnancy and Childbirth Group) (January 2024). "Vitamin B12 supplementation during pregnancy for maternal and child health outcomes". The Cochrane Database of Systematic Reviews. 1 (1): CD013823. doi:10.1002/14651858.CD013823.pub2. PMC 10772977. PMID 38189492.
- ^ a b Stabler SP (2020). "Vitamin B12". In Marriott BP, Birt DF, Stallings VA, Yates AA (eds.). Present Knowledge in Nutrition, Eleventh Edition. London: Academic Press (Elsevier). pp. 257–272. ISBN 978-0-323-66162-1.
US survey data from the NHANES What We Eat in America 2013e16 cohort reported the median vitamin B12 consumption for all adult men of 5.1 mcg and women of 3.5 mcg.95b Using the Estimated Average Requirement (EAR) for adults for Vitamin B12 of 2 mcg,93 less than 3% of men and 8% of women in the United States had inadequate diets using this comparator. However, 11% of girls 14e18 years had intakes less than their EAR of 2.0 mcg.
- ^ a b c d e f g h i j k l Institute of Medicine (1998). "Vitamin B12". Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: The National Academies Press. pp. 306–356. ISBN 978-0-309-06554-2. Retrieved 7 February 2012.
- ^ Choudhury A, Jena A, Jearth V, Dutta AK, Makharia G, Dutta U, et al. (May 2023). "Vitamin B12 deficiency and use of proton pump inhibitors: a systematic review and meta-analysis". Expert Rev Gastroenterol Hepatol. 17 (5): 479–487. doi:10.1080/17474124.2023.2204229. PMID 37060552.
- ^ "Acid-Reflux Drugs Tied to Lower Levels of Vitamin B-12". WebMD. Archived from the original on 23 July 2018. Retrieved 23 July 2018.
- ^ a b c "Vitamin B12 Deficiency Anemia". www.hopkinsmedicine.org. 8 August 2021. Retrieved 16 February 2022.
- ^ "Pernicious anemia: MedlinePlus Medical Encyclopedia". medlineplus.gov. Retrieved 6 January 2022.
- ^ Baik HW, Russell RM (18 November 2021). "Vitamin B12 deficiency in the elderly". Annual Review of Nutrition. 19: 357–377. doi:10.1146/annurev.nutr.19.1.357. PMID 10448529.
- ^ Butler P, Kräutler B (2006). "Biological Organometallic Chemistry of B12". Bioorganometallic Chemistry. Topics in Organometallic Chemistry. Vol. 17. pp. 1–55. doi:10.1007/3418_004. ISBN 3-540-33047-X.
- ^ a b Banerjee R, Ragsdale SW (July 2003). "The many faces of vitamin B12: catalysis by cobalamin-dependent enzymes". Annual Review of Biochemistry. 72: 209–247. doi:10.1146/annurev.biochem.72.121801.161828. PMID 14527323. S2CID 37393683.
- ^ Obeid R, Fedosov SN, Nexo E (July 2015). "Cobalamin coenzyme forms are not likely to be superior to cyano- and hydroxyl-cobalamin in prevention or treatment of cobalamin deficiency". Molecular Nutrition & Food Research. 59 (7): 1364–1372. doi:10.1002/mnfr.201500019. PMC 4692085. PMID 25820384.
- ^ Paul C, Brady DM (February 2017). "Comparative Bioavailability and Utilization of Particular Forms of B12 Supplements With Potential to Mitigate B12-related Genetic Polymorphisms". Integr Med (Encinitas). 16 (1): 42–49. PMC 5312744. PMID 28223907.
- ^ a b c d Watanabe F, Bito T (September 2018). "Determination of Cobalamin and Related Compounds in Foods". J AOAC Int. 101 (5): 1308–1313. doi:10.5740/jaoacint.18-0045. PMID 29669618. S2CID 4978703.
- ^ a b c Watanabe F, Katsura H, Takenaka S, Fujita T, Abe K, Tamura Y, et al. (November 1999). "Pseudovitamin B(12) is the predominant cobamide of an algal health food, spirulina tablets". J. Agric. Food Chem. 47 (11): 4736–4741. doi:10.1021/jf990541b. PMID 10552882.
- ^ van der Put NM, van Straaten HW, Trijbels FJ, Blom HJ (April 2001). "Folate, homocysteine and neural tube defects: an overview". Experimental Biology and Medicine. 226 (4): 243–270. doi:10.1177/153537020122600402. PMID 11368417. S2CID 29053617.
- ^ Skerrett PJ (February 2019). "Vitamin B12 deficiency can be sneaky, harmful". Harvard Health Blog. Archived from the original on 29 October 2019. Retrieved 6 January 2020.
- ^ "Vitamin B12 or folate deficiency anaemia – Symptoms". National Health Service, England. 23 May 2019. Archived from the original on 12 August 2017. Retrieved 6 January 2020.
- ^ Masalha R, Chudakov B, Muhamad M, Rudoy I, Volkov I, Wirguin I (September 2001). "Cobalamin-responsive psychosis as the sole manifestation of vitamin B12 deficiency". The Israel Medical Association Journal. 3 (9): 701–703. PMID 11574992.
- ^ a b Lachner C, Steinle NI, Regenold WT (2012). "The neuropsychiatry of vitamin B12 deficiency in elderly patients". J Neuropsychiatry Clin Neurosci. 24 (1): 5–15. doi:10.1176/appi.neuropsych.11020052. PMID 22450609. S2CID 20350330.
- ^ Bennett M (March 2001). "Vitamin B12 deficiency, infertility and recurrent fetal loss". The Journal of Reproductive Medicine. 46 (3): 209–212. PMID 11304860.
- ^ "What Is Pernicious Anemia?". NHLBI. 1 April 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ Briani C, Dalla Torre C, Citton V, Manara R, Pompanin S, Binotto G, et al. (November 2013). "Cobalamin Deficiency: Clinical Picture and Radiological Findings". Nutrients. 5 (11): 4521–4539. doi:10.3390/nu5114521. ISSN 2072-6643. PMC 3847746. PMID 24248213.
- ^ Amarapurka DN, Patel ND (September 2004). "Gastric Antral Vascular Ectasia (GAVE) Syndrome" (PDF). Journal of the Association of Physicians of India. 52: 757. Archived (PDF) from the original on 4 March 2016.
- ^ Greenburg M (2010). Handbook of Neurosurgery 7th Edition. New York: Thieme Publishers. pp. 1187–1188. ISBN 978-1-60406-326-4.
- ^ Lerner NB (2016). "Vitamin B12 Deficiency". In Kliegman RM, Stanton B, St Geme J, Schor NF (eds.). Nelson Textbook of Pediatrics (20th ed.). Elsevier Health Sciences. pp. 2319–2326. ISBN 978-1-4557-7566-8.
- ^ a b Melina V, Craig W, Levin S (December 2016). "Position of the Academy of Nutrition and Dietetics: Vegetarian Diets". Journal of the Academy of Nutrition and Dietetics. 116 (12): 1970–1980. doi:10.1016/j.jand.2016.09.025. PMID 27886704. S2CID 4984228.
Fermented foods (such as tempeh), nori, spirulina, chlorella algae, and unfortified nutritional yeast cannot be relied upon as adequate or practical sources of B-12.39,40 Vegans must regularly consume reliable sources—meaning B-12 fortified foods or B-12 containing supplements—or they could become deficient, as shown in case studies of vegan infants, children, and adults.
- ^ Pawlak R, Parrott SJ, Raj S, Cullum-Dugan D, Lucus D (February 2013). "How prevalent is vitamin B(12) deficiency among vegetarians?". Nutrition Reviews. 71 (2): 110–117. doi:10.1111/nure.12001. PMID 23356638.
- ^ a b Woo KS, Kwok TC, Celermajer DS (August 2014). "Vegan diet, subnormal vitamin B-12 status and cardiovascular health". Nutrients. 6 (8): 3259–3273. doi:10.3390/nu6083259. PMC 4145307. PMID 25195560.
- ^ Stockton L, Simonsen C, Seago S (2017). "Nitrous oxide–induced vitamin B12 deficiency". Proceedings (Baylor University. Medical Center). 30 (2): 171–172. doi:10.1080/08998280.2017.11929571. PMC 5349816. PMID 28405070.
- ^ a b c d e f g h i j Obeid R, Murphy M, Solé-Navais P, Yajnik C (November 2017). "Cobalamin Status from Pregnancy to Early Childhood: Lessons from Global Experience". Advances in Nutrition. 8 (6): 971–979. doi:10.3945/an.117.015628. PMC 5683008. PMID 29141978.
- ^ a b "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017. Archived (PDF) from the original on 7 January 2020. Retrieved 28 August 2017.
- ^ Rogne T, Tielemans MJ, Chong MF, Yajnik CS, Krishnaveni GV, Poston L, et al. (February 2017). "Associations of Maternal Vitamin B12 Concentration in Pregnancy With the Risks of Preterm Birth and Low Birth Weight: A Systematic Review and Meta-Analysis of Individual Participant Data". Am J Epidemiol. 185 (3): 212–223. doi:10.1093/aje/kww212. PMC 5390862. PMID 28108470.
- ^ a b Sebastiani G, Herranz Barbero A, Borrás-Novell C, Alsina Casanova M, Aldecoa-Bilbao V, Andreu-Fernández V, et al. (March 2019). "The Effects of Vegetarian and Vegan Diet during Pregnancy on the Health of Mothers and Offspring". Nutrients. 11 (3): 557. doi:10.3390/nu11030557. PMC 6470702. PMID 30845641.
- ^ Wilson RD, O'Connor DL (June 2022). "Guideline No. 427: Folic Acid and Multivitamin Supplementation for Prevention of Folic Acid-Sensitive Congenital Anomalies". Journal of Obstetrics and Gynaecology Canada. 44 (6): 707–719.e1. doi:10.1016/j.jogc.2022.04.004. PMID 35691683.
- ^ "Nutrition During Pregnancy". www.acog.org. Retrieved 15 January 2024.
- ^ "Pregnancy: Vegetarian Diet". myhealth.alberta.ca. Retrieved 15 January 2024.
- ^ a b "Map: Count of Nutrients In Fortification Standards". Global Fortification Data Exchange. Archived from the original on 11 April 2019. Retrieved 15 April 2020.
- ^ Weng TC, Chang CH, Dong YH, Chang YC, Chuang LM (July 2015). "Anaemia and related nutrient deficiencies after Roux-en-Y gastric bypass surgery: a systematic review and meta-analysis". BMJ Open. 5 (7): e006964. doi:10.1136/bmjopen-2014-006964. PMC 4513480. PMID 26185175.
- ^ Majumder S, Soriano J, Louie Cruz A, Dasanu CA (2013). "Vitamin B12 deficiency in patients undergoing bariatric surgery: preventive strategies and key recommendations". Surg Obes Relat Dis. 9 (6): 1013–1019. doi:10.1016/j.soard.2013.04.017. PMID 24091055.
- ^ a b Mahawar KK, Reid A, Graham Y, Callejas-Diaz L, Parmar C, Carr WR, et al. (July 2018). "Oral Vitamin B12 Supplementation After Roux-en-Y Gastric Bypass: a Systematic Review". Obes Surg. 28 (7): 1916–1923. doi:10.1007/s11695-017-3102-y. PMID 29318504. S2CID 35209784.
- ^ Shipton MJ, Thachil J (April 2015). "Vitamin B12 deficiency – A 21st century perspective". Clin Med (Lond). 15 (2): 145–150. doi:10.7861/clinmedicine.15-2-145. PMC 4953733. PMID 25824066.
- ^ Devalia V, Hamilton MS, Molloy AM (August 2014). "Guidelines for the diagnosis and treatment of cobalamin and folate disorders". Br. J. Haematol. 166 (4): 496–513. doi:10.1111/bjh.12959. PMID 24942828. S2CID 5772424.
- ^ Moretti R, Caruso P (January 2019). "The Controversial Role of Homocysteine in Neurology: From Labs to Clinical Practice". Int J Mol Sci. 20 (1): 231. doi:10.3390/ijms20010231. PMC 6337226. PMID 30626145.
- ^ "Methylmalonic acidemia". Genetics Home Reference. US National Library of Medicine. October 2015. Retrieved 10 July 2022.
- ^ Devalia V (August 2006). "Diagnosing vitamin B-12 deficiency on the basis of serum B-12 assay". BMJ. 333 (7564): 385–386. doi:10.1136/bmj.333.7564.385. PMC 1550477. PMID 16916826.
- ^ McCorvie TJ, Ferreira D, Yue WW, Froese DS (May 2023). "The complex machinery of human cobalamin metabolism". J Inherit Metab Dis. 46 (3): 406–20. doi:10.1002/jimd.12593. PMID 36680553.
- ^ Ermens AA, Vlasveld LT, Lindemans J (November 2003). "Significance of elevated cobalamin (vitamin B12) levels in blood". Clin Biochem. 36 (8): 585–90. doi:10.1016/j.clinbiochem.2003.08.004. PMID 14636871.
- ^ a b Elangovan R, Baruteau J (September 2022). "Inherited and acquired vitamin B12 deficiencies: Which administration route to choose for supplementation?". Front Pharmacol. 13: 972468. doi:10.3389/fphar.2022.972468. PMC 9559827. PMID 36249776.
- ^ a b Andrès E, Zulfiqar AA, Serraj K, Vogel T, Kaltenbach G (September 2018). "Systematic review and pragmatic clinical approach to oral and nasal vitamin B12 (cobalamin) treatment in patients with vitamin B12 deficiency related to gastrointestinal disorders". J Clin Med. 7 (10): 304–17. doi:10.3390/jcm7100304. PMC 6210286. PMID 30261596.
- ^ Hall AH, Rumack BH (1987). "Hydroxycobalamin/sodium thiosulfate as a cyanide antidote". The Journal of Emergency Medicine. 5 (2): 115–121. doi:10.1016/0736-4679(87)90074-6. PMID 3295013.
- ^ MacLennan L, Moiemen N (February 2015). "Management of cyanide toxicity in patients with burns". Burns. 41 (1): 18–24. doi:10.1016/j.burns.2014.06.001. PMID 24994676.
- ^ Dart RC (2006). "Hydroxocobalamin for acute cyanide poisoning: new data from preclinical and clinical studies; new results from the prehospital emergency setting". Clinical Toxicology. 44 (Suppl. 1): 1–3. doi:10.1080/15563650600811607. PMID 16990188.
- ^ a b c "Foods highest in Vitamin B12 (based on levels per 100-gram serving)". Nutrition Data. Condé Nast, USDA National Nutrient Database, release SR-21. 2014. Archived from the original on 16 November 2019. Retrieved 16 February 2017.
- ^ "Tolerable Upper Intake Levels For Vitamins And Minerals" (PDF). European Food Safety Authority. 2006. Archived (PDF) from the original on 15 October 2019. Retrieved 12 March 2016.
- ^ "Dietary Reference Intakes for Japanese 2010: Water-Soluble Vitamins" Journal of Nutritional Science and Vitaminology 2013(59):S67–S82.
- ^ World Health Organization (2005). "Chapter 14: Vitamin B12". Vitamin and Mineral Requirements in Human Nutrition (2nd ed.). Geneva: World Health Organization. pp. 279–287. hdl:10665/42716. ISBN 978-92-4-154612-6.
- ^ "Food Labeling: Revision of the Nutrition and Supplement Facts Labels" (PDF). Federal Register. 27 May 2016. p. 33982. Archived (PDF) from the original on 8 August 2016. Retrieved 27 August 2017.
- ^ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". Dietary Supplement Label Database (DSLD). Archived from the original on 7 April 2020. Retrieved 16 May 2020.
- ^ "Changes to the Nutrition Facts Label". U.S. Food and Drug Administration (FDA). 27 May 2016. Retrieved 16 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Industry Resources on the Changes to the Nutrition Facts Label". U.S. Food and Drug Administration (FDA). 21 December 2018. Retrieved 16 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Fang H, Kang J, Zhang D (January 2017). "Microbial production of vitamin B12: a review and future perspectives". Microbial Cell Factories. 16 (1): 15. doi:10.1186/s12934-017-0631-y. PMC 5282855. PMID 28137297.
- ^ Moore SJ, Warren MJ (June 2012). "The anaerobic biosynthesis of vitamin B12" (PDF). Biochemical Society Transactions. 40 (3): 581–586. doi:10.1042/BST20120066. PMID 22616870. S2CID 26057998.
- ^ Graham RM, Deery E, Warren MJ (2009). "18: Vitamin B12: Biosynthesis of the Corrin Ring". In Warren MJ, Smith (eds.). Tetrapyrroles Birth, Life and Death. New York: Springer-Verlag. p. 286. doi:10.1007/978-0-387-78518-9_18. ISBN 978-0-387-78518-9.
- ^ a b Gille D, Schmid A (February 2015). "Vitamin B12 in meat and dairy products". Nutrition Reviews. 73 (2): 106–115. doi:10.1093/nutrit/nuu011. PMID 26024497.
- ^ a b c Stevens CE, Hume ID (April 1998). "Contributions of microbes in vertebrate gastrointestinal tract to production and conservation of nutrients". Physiol. Rev. 78 (2): 393–427. doi:10.1152/physrev.1998.78.2.393. PMID 9562034. S2CID 103191.
- ^ McDowell LR (2008). Vitamins in Animal and Human Nutrition (2nd ed.). Hoboken: John Wiley & Sons. pp. 525, 539. ISBN 978-0-470-37668-3. Archived from the original on 8 September 2017. Retrieved 17 January 2017.
- ^ Erickson A (3 September 2019). "Cobalt deficiency in sheep and cattle". Department of Primary Industries and Regional Development. Government of Western Australia. Archived from the original on 11 November 2015. Retrieved 18 April 2020.
- ^ a b Rooke J (30 October 2013). "Do carnivores need Vitamin B12 supplements?". Baltimore Post Examiner. Archived from the original on 16 January 2017. Retrieved 17 January 2017.
- ^ a b c d Watanabe F (November 2007). "Vitamin B12 sources and bioavailability". Experimental Biology and Medicine. 232 (10): 1266–1274. doi:10.3181/0703-MR-67. PMID 17959839. S2CID 14732788.
- ^ Dossey AT (1 February 2013). "Why Insects Should Be in Your Diet". The Scientist. Archived from the original on 11 November 2017. Retrieved 18 April 2020.
- ^ a b "Vitamin B-12 (μg)" (PDF). USDA National Nutrient Database for Standard Reference Release 28. 27 October 2015. Archived (PDF) from the original on 26 January 2017. Retrieved 6 January 2020.
- ^ a b Keuth S, Bisping B (May 1994). "Vitamin B12 production by Citrobacter freundii or Klebsiella pneumoniae during tempeh fermentation and proof of enterotoxin absence by PCR". Applied and Environmental Microbiology. 60 (5): 1495–1499. Bibcode:1994ApEnM..60.1495K. doi:10.1128/AEM.60.5.1495-1499.1994. PMC 201508. PMID 8017933.
- ^ Mo H, Kariluoto S, Piironen V, Zhu Y, Sanders MG, Vincken JP, et al. (December 2013). "Effect of soybean processing on content and bioaccessibility of folate, vitamin B12 and isoflavones in tofu and tempe". Food Chemistry. 141 (3): 2418–2425. doi:10.1016/j.foodchem.2013.05.017. PMID 23870976.
- ^ Watanabe F, Yabuta Y, Bito T, Teng F (May 2014). "Vitamin B₁₂-containing plant food sources for vegetarians". Nutrients. 6 (5): 1861–1873. doi:10.3390/nu6051861. PMC 4042564. PMID 24803097.
- ^ Kwak CS, Lee MS, Lee HJ, Whang JY, Park SC (June 2010). "Dietary source of vitamin B(12) intake and vitamin B(12) status in female elderly Koreans aged 85 and older living in rural area". Nutrition Research and Practice. 4 (3): 229–234. doi:10.4162/nrp.2010.4.3.229. PMC 2895704. PMID 20607069.
- ^ Kwak CS, Lee MS, Oh SI, Park SC (2010). "Discovery of novel sources of vitamin b(12) in traditional korean foods from nutritional surveys of centenarians". Current Gerontology and Geriatrics Research. 2010: 374897. doi:10.1155/2010/374897. PMC 3062981. PMID 21436999.
- ^ Kumudha A, Selvakumar S, Dilshad P, Vaidyanathan G, Thakur MS, Sarada R (March 2015). "Methylcobalamin – a form of vitamin B12 identified and characterised in Chlorella vulgaris". Food Chemistry. 170: 316–320. doi:10.1016/j.foodchem.2014.08.035. PMID 25306351.
- ^ Martins JH, Barg H, Warren MJ, Jahn D (March 2002). "Microbial production of vitamin B12". Appl Microbiol Biotechnol. 58 (3): 275–285. doi:10.1007/s00253-001-0902-7. PMID 11935176. S2CID 22232461.
- ^ Mangels R. "Vitamin B12 in the Vegan Diet". Vegetarian Resource Group. Archived from the original on 19 December 2012. Retrieved 17 January 2008.
- ^ "Don't Vegetarians Have Trouble Getting Enough Vitamin B12?". Physicians Committee for Responsible Medicine. Archived from the original on 8 October 2011. Retrieved 17 January 2008.
- ^ a b European Food Safety Authority (25 September 2008). "5′-deoxyadenosylcobalamin and methylcobalamin as sources for Vitamin B12 added as a nutritional substance in food supplements: Scientific opinion of the Scientific Panel on Food Additives and Nutrient Sources added to food". EFSA Journal. 815 (10): 1–21. doi:10.2903/j.efsa.2008.815. "the metabolic fate and biological distribution of methylcobalamin and 5′-deoxyadenosylcobalamin are expected to be similar to that of other sources of vitamin B12 in the diet".
- ^ Thakkar K, Billa G (January 2015). "Treatment of vitamin B12 deficiency – Methylcobalamine? Cyancobalamine? Hydroxocobalamin? – clearing the confusion". European Journal of Clinical Nutrition. 69 (1): 1–2. doi:10.1038/ejcn.2014.165. PMID 25117994.
- ^ Obeid R, Fedosov SN, Nexo E (28 March 2015). "Cobalamin coenzyme forms are not likely to be superior to cyano- and hydroxyl-cobalamin in prevention or treatment of cobalamin deficiency". Molecular Nutrition & Food Research. 59 (7): 1364–1372. doi:10.1002/mnfr.201500019. PMC 4692085. PMID 25820384.
- ^ Obeid R, Andrès E, Češka R, Hooshmand B, Guéant-Rodriguez R, Prada GI, et al. (10 April 2024). "Diagnosis, Treatment and Long-Term Management of Vitamin B12 Deficiency in Adults: A Delphi Expert Consensus". Journal of Clinical Medicine. 13 (8): 2176. doi:10.3390/jcm13082176. PMID 38673453.
- ^ Lane LA, Rojas-Fernandez C (July–August 2002). "Treatment of vitamin b(12)-deficiency anemia: oral versus parenteral therapy". The Annals of Pharmacotherapy. 36 (7–8): 1268–1272. doi:10.1345/aph.1A122. PMID 12086562. S2CID 919401.
- ^ Butler CC, Vidal-Alaball J, Cannings-John R, McCaddon A, Hood K, Papaioannou A, et al. (June 2006). "Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency: a systematic review of randomized controlled trials". Family Practice. 23 (3): 279–285. CiteSeerX 10.1.1.488.7931. doi:10.1093/fampra/cml008. PMID 16585128.
- ^ Arslan SA, Arslan I, Tirnaksiz F (March 2013). "Cobalamins and Methylcobalamin: Coenzyme of Vitamin B12". FABAD J. Pharm. Sci. 38 (3): 151–157. S2CID 1929961.
- ^ Wang C, Li D, Cai F, Zhang X, Xu X, Liu X, et al. (October 2019). "Mutation spectrum of MMACHC in Chinese pediatric patients with cobalamin C disease: A case series and literature review". Eur J Med Genet. 62 (10): 103713. doi:10.1016/j.ejmg.2019.103713. PMID 31279840.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Thauvin-Robinet C, Roze E, Couvreur G, Horellou MH, Sedel F, Grabli D, et al. (June 2008). "The adolescent and adult form of cobalamin C disease: clinical and molecular spectrum". Journal of Neurology, Neurosurgery, and Psychiatry. 79 (6): 725–28. doi:10.1136/jnnp.2007.133025. PMID 18245139. S2CID 23493993.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Fidaleo M, Tacconi S, Sbarigia C, Passeri D, Rossi M, Tata AM, et al. (March 2021). "Current Nanocarrier Strategies Improve Vitamin B12 Pharmacokinetics, Ameliorate Patients' Lives, and Reduce Costs". Nanomaterials. 11 (3): 743. doi:10.3390/nano11030743. PMC 8001893. PMID 33809596.
- ^ Yamada K, Yamada Y, Fukuda M, Yamada S (November 1999). "Bioavailability of Dried Asakusanori (Porphyra tenera) as a Source of Cobalamin (Vitamin B12)". International Journal for Vitamin and Nutrition Research. 69 (6): 412–418. doi:10.1024/0300-9831.69.6.412. PMID 10642899.
- ^ Schmidt A, Call LM, Macheiner L, Mayer HK (May 2019). "Determination of vitamin B12 in four edible insect species by immunoaffinity and ultra-high performance liquid chromatography". Food Chemistry. 281: 124–129. doi:10.1016/j.foodchem.2018.12.039. PMID 30658738. S2CID 58651702.
- ^ Yamada K, Shimodaira M, Chida S, Yamada N, Matsushima N, Fukuda M, et al. (2008). "Degradation of vitamin B12 in dietary supplements". International Journal for Vitamin and Nutrition Research. 78 (4–5): 195–203. doi:10.1024/0300-9831.78.45.195. PMID 19326342.
- ^ Kräutler B (December 2020). "Antivitamins B12 -Some Inaugural Milestones". Chemistry: A European Journal. 26 (67): 15438–15445. doi:10.1002/chem.202003788. PMC 7756841. PMID 32956545.
- ^ DeVault KR, Talley NJ (September 2009). "Insights into the future of gastric acid suppression". Nat Rev Gastroenterol Hepatol. 6 (9): 524–532. doi:10.1038/nrgastro.2009.125. PMID 19713987. S2CID 25413839.
- ^ Ahmed MA (2016). "Metformin and Vitamin B12 Deficiency: Where Do We Stand?". Journal of Pharmacy & Pharmaceutical Sciences. 19 (3): 382–398. doi:10.18433/J3PK7P. hdl:2263/60716. PMID 27806244.
- ^ Gilligan MA (February 2002). "Metformin and vitamin B12 deficiency". Archives of Internal Medicine. 162 (4): 484–485. doi:10.1001/archinte.162.4.484. PMID 11863489.
- ^ Copp S (1 December 2007). "What effect does metformin have on vitamin B12 levels?". UK Medicines Information, NHS. Archived from the original on 27 September 2007.
- ^ a b "Vitamin B-12: Interactions". WebMD. Retrieved 21 April 2020.
- ^ Linnebank M, Moskau S, Semmler A, Widman G, Stoffel-Wagner B, Weller M, et al. (February 2011). "Antiepileptic drugs interact with folate and vitamin B12 serum levels" (PDF). Ann. Neurol. 69 (2): 352–359. doi:10.1002/ana.22229. PMID 21246600. S2CID 7282489.
- ^ Giedyk M, Goliszewska K, Gryko D (June 2015). "Vitamin B12 catalysed reactions". Chemical Society Reviews. 44 (11): 3391–3404. doi:10.1039/C5CS00165J. PMID 25945462.
- ^ Jaouen G, ed. (2006). Bioorganometallics: Biomolecules, Labeling, Medicine. Weinheim: Wiley-VCH. pp. 17–25. ISBN 978-3-527-30990-0.
- ^ a b Lawrance P (March 2015). "Vitamin B12: A review of analytical methods for use in food". LGC Limited.
- ^ O'Leary F, Samman S (March 2010). "Vitamin B12 in Health and Disease". Nutrients. 2 (3): 299–316. doi:10.3390/nu2030299. ISSN 2072-6643. PMC 3257642. PMID 22254022.
- ^ Obeid R, ed. (12 July 2017). Vitamin B12. CRC Press. doi:10.1201/9781315119540. ISBN 978-1-315-11954-0.
- ^ a b Takahashi-Iñiguez T, García-Hernandez E, Arreguín-Espinosa R, Flores ME (June 2012). "Role of vitamin B12 on methylmalonyl-CoA mutase activity". J Zhejiang Univ Sci B. 13 (6): 423–437. doi:10.1631/jzus.B1100329. PMC 3370288. PMID 22661206.
- ^ a b c Froese DS, Fowler B, Baumgartner MR (July 2019). "Vitamin B12, folate, and the methionine remethylation cycle-biochemistry, pathways, and regulation" (PDF). Journal of Inherited Metabolic Disease. 42 (4): 673–685. doi:10.1002/jimd.12009. PMID 30693532.
- ^ Reinhold A, Westermann M, Seifert J, von Bergen M, Schubert T, Diekert G (November 2012). "Impact of vitamin B12 on formation of the tetrachloroethene reductive dehalogenase in Desulfitobacterium hafniense strain Y51". Appl. Environ. Microbiol. 78 (22): 8025–8032. Bibcode:2012ApEnM..78.8025R. doi:10.1128/AEM.02173-12. PMC 3485949. PMID 22961902.
- ^ Payne KA, Quezada CP, Fisher K, Dunstan MS, Collins FA, Sjuts H, et al. (January 2015). "Reductive dehalogenase structure suggests a mechanism for B12-dependent dehalogenation". Nature. 517 (7535): 513–516. Bibcode:2015Natur.517..513P. doi:10.1038/nature13901. PMC 4968649. PMID 25327251.
- ^ Ballhausen D, Mittaz L, Boulat O, Bonafé L, Braissant O (December 2009). "Evidence for catabolic pathway of propionate metabolism in CNS: expression pattern of methylmalonyl-CoA mutase and propionyl-CoA carboxylase alpha-subunit in developing and adult rat brain". Neuroscience. 164 (2): 578–587. doi:10.1016/j.neuroscience.2009.08.028. PMID 19699272. S2CID 34612963.
- ^ Marsh EN (1999). "Coenzyme B12 (cobalamin)-dependent enzymes". Essays Biochem. 34: 139–154. doi:10.1042/bse0340139. PMID 10730193.
- ^ a b c d e Guéant JL, Guéant-Rodriguez RM, Alpers DH (2022). "Vitamin B12 absorption and malabsorption". Vitam Horm. Vitamins and Hormones. 119: 241–274. doi:10.1016/bs.vh.2022.01.016. ISBN 978-0-323-99223-7. PMID 35337622.
- ^ Maton A, Hopkins J, McLaughlin CW, Johnson S, Warner MQ, LaHart D, et al. (1993). Human Biology and Health. Englewood Cliffs, New Jersey, USA: Prentice Hall. ISBN 978-0-13-981176-0.
- ^ Seetharam B, Li N (2000). "Transcobalamin II and its cell surface receptor". Vitam Horm. Vitamins & Hormones. 59: 337–66. doi:10.1016/s0083-6729(00)59012-8. ISBN 978-0-12-709859-3. PMID 10714245.
- ^ Lam JR, Schneider JL, Zhao W, Corley DA (December 2013). "Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency". JAMA. 310 (22): 2435–42. doi:10.1001/jama.2013.280490. PMID 24327038.
- ^ Baik HW, Russell RM (1999). "Vitamin B12 deficiency in the elderly". Annual Review of Nutrition. 19: 357–77. doi:10.1146/annurev.nutr.19.1.357. PMID 10448529.
- ^ "Vitamin B12 Deficiency – Nutritional Disorders". MSD Manual Professional Edition. Retrieved 24 May 2022.
- ^ Kovatcheva M, Melendez E, Chondronasiou D, Pietrocola F, Bernad R, Caballe A, et al. (November 2023). "Vitamin B12 is a limiting factor for induced cellular plasticity and tissue repair". Nature Metabolism. 5 (11): 1911–1930. doi:10.1038/s42255-023-00916-6. PMC 10663163. PMID 37973897.
- ^ Vílchez-Acosta A, Desdín-Micó G, Ocampo A (November 2023). "Vitamin B12 emerges as key player during cellular reprogramming". Nature Metabolism. 5 (11): 1844–1845. doi:10.1038/s42255-023-00917-5. PMID 37973898. S2CID 265273574.
- ^ Battersby AR, Fookes CJ, Matcham GW, McDonald E (May 1980). "Biosynthesis of the pigments of life: formation of the macrocycle". Nature. 285 (5759): 17–21. Bibcode:1980Natur.285...17B. doi:10.1038/285017a0. PMID 6769048. S2CID 9070849.
- ^ Frank S, Brindley AA, Deery E, Heathcote P, Lawrence AD, Leech HK, et al. (August 2005). "Anaerobic synthesis of vitamin B12: characterization of the early steps in the pathway" (PDF). Biochemical Society Transactions. 33 (Pt 4): 811–814. doi:10.1042/BST0330811. PMID 16042604.
- ^ Battersby AR (1993). "How Nature builds the pigments of life" (PDF). Pure and Applied Chemistry. 65 (6): 1113–1122. doi:10.1351/pac199365061113. S2CID 83942303. Archived (PDF) from the original on 24 July 2018. Retrieved 20 February 2020.
- ^ Battersby A (2005). "Chapter 11: Discovering the wonder of how Nature builds its molecules". In Archer MD, Haley CD (eds.). The 1702 chair of chemistry at Cambridge: transformation and change. Cambridge University Press. pp. xvi, 257–282. ISBN 0-521-82873-2.
- ^ Perlman D (1959). "Microbial synthesis of cobamides". Advances in Applied Microbiology. 1: 87–122. doi:10.1016/S0065-2164(08)70476-3. ISBN 978-0-12-002601-2. PMID 13854292.
- ^ Martens JH, Barg H, Warren MJ, Jahn D (March 2002). "Microbial production of vitamin B12". Applied Microbiology and Biotechnology. 58 (3): 275–285. doi:10.1007/s00253-001-0902-7. PMID 11935176. S2CID 22232461.
- ^ a b c Fang H, Kang J, Zhang D (January 2017). "Microbial production of vitamin B12: a review and future perspectives". Microb. Cell Fact. 16 (1): 15. doi:10.1186/s12934-017-0631-y. PMC 5282855. PMID 28137297.
- ^ Linnell JC, Matthews DM (February 1984). "Cobalamin metabolism and its clinical aspects" (PDF). Clinical Science. 66 (2): 113–121. doi:10.1042/cs0660113. PMID 6420106. S2CID 27191837.
- ^ Piwowarek K, Lipińska E, Hać-Szymańczuk E, Kieliszek M, Ścibisz I (January 2018). "Propionibacterium spp.-source of propionic acid, vitamin B12, and other metabolites important for the industry". Appl. Microbiol. Biotechnol. 102 (2): 515–538. doi:10.1007/s00253-017-8616-7. PMC 5756557. PMID 29167919.
- ^ Riaz M, Ansari ZA, Iqbal F, Akram M (2007). "Microbial production of vitamin B12 by methanol utilizing strain of Pseudomonas species". Pakistan Journal of Biochemistry & Molecular Biology. 40: 5–10.[permanent dead link]
- ^ Zhang Y (26 January 2009). "New round of price slashing in vitamin B12 sector (Fine and Specialty)". China Chemical Reporter. Archived from the original on 13 May 2013.
- ^ Khan AG, Eswaran SV (June 2003). "Woodward's synthesis of vitamin B12". Resonance. 8 (6): 8–16. doi:10.1007/BF02837864. S2CID 120110443.
- ^ Eschenmoser A, Wintner CE (June 1977). "Natural product synthesis and vitamin B12". Science. 196 (4297): 1410–1420. Bibcode:1977Sci...196.1410E. doi:10.1126/science.867037. PMID 867037.
- ^ a b Riether D, Mulzer J (2003). "Total Synthesis of Cobyric Acid: Historical Development and Recent Synthetic Innovations". European Journal of Organic Chemistry. 2003: 30–45. doi:10.1002/1099-0690(200301)2003:1<30::AID-EJOC30>3.0.CO;2-I.
- ^ a b "Synthesis of Cyanocobalamin by Robert B. Woodward (1973)". www.synarchive.com. Archived from the original on 16 February 2018. Retrieved 15 February 2018.
- ^ a b c Greer JP (2014). Wintrobe's Clinical Hematology Thirteenth Edition. Philadelphia, PA: Wolters Kluwer/Lippincott Williams & Wilkins. ISBN 978-1-4511-7268-3. Chapter 36: Megaloblastic anemias: disorders of impaired DNA synthesis by Ralph Carmel
- ^ "George H. Whipple – Biographical". www.nobelprize.org. Archived from the original on 13 September 2017. Retrieved 10 October 2017.
- ^ "The Nobel Prize in Physiology or Medicine 1934". NobelPrize.org. Retrieved 23 February 2023.
- ^ "Mary Shorb Lecture in Nutrition". Archived from the original on 4 March 2016. Retrieved 3 March 2016.
- ^ Shorb MS (May 10, 2012). "Annual Lecture". Department of Animal & Avian Sciences, University of Maryland. Archived from the original on December 12, 2012. Retrieved August 2, 2014.
- ^ Hodgkin DC, Pickworth J, Robertson JH, Trueblood KN, Prosen RJ, White JG (1955). "Structure of Vitamin B12: The Crystal Structure of the Hexacarboxylic Acid derived from B12 and the Molecular Structure of the Vitamin". Nature. 176 (4477): 325–28. Bibcode:1955Natur.176..325H. doi:10.1038/176325a0. PMID 13253565. S2CID 4220926.
- ^ Hodgkin DC, Kamper J, Mackay M, Pickworth J, Trueblood KN, White JG (July 1956). "Structure of vitamin B12". Nature. 178 (4524): 64–66. Bibcode:1956Natur.178...64H. doi:10.1038/178064a0 (inactive 3 December 2024). PMID 13348621. S2CID 4210164.
{{cite journal}}: CS1 maint: DOI inactive as of December 2024 (link) - ^ a b Dodson G (December 2002). "Dorothy Mary Crowfoot Hodgkin, 12 May 1910 – 29 July 1994". Biographical Memoirs of Fellows of the Royal Society. 48: 181–219. doi:10.1098/rsbm.2002.0011. PMID 13678070. S2CID 61764553.
- ^ Carpenter KJ. "The Nobel Prize and the discovery of vitamins". nobelprize.org. Archived from the original on 20 August 2023. Retrieved 19 November 2023.
- ^ a b Gaby AR (October 2002). "Intravenous nutrient therapy: the "Myers' cocktail"". Altern Med Rev. 7 (5): 389–403. PMID 12410623.
- ^ Gavura S (24 May 2013). "A closer look at vitamin injections". Science-Based Medicine. Archived from the original on 11 January 2020. Retrieved 10 January 2020.
- ^ Bauer BA (29 March 2018). "Are vitamin B-12 injections helpful for weight loss?". Mayo Clinic. Archived from the original on 27 November 2019. Retrieved 11 January 2020.
- ^ Silverstein WK, Lin Y, Dharma C, Croxford R, Earle CC, Cheung MC (July 2019). "Prevalence of Inappropriateness of Parenteral Vitamin B12 Administration in Ontario, Canada". JAMA Internal Medicine. 179 (10): 1434–1436. doi:10.1001/jamainternmed.2019.1859. ISSN 2168-6106. PMC 6632124. PMID 31305876.
External links
- Cyanocobalamin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)






