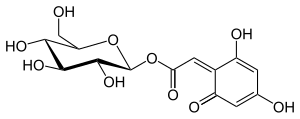
Anthocyanone A
| |
| Names | |
|---|---|
| IUPAC name [(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] 2-(2,6-dihydroxy-4-oxocyclohexa-2,5-dien-1-ylidene)acetate | |
| Other names 8-β-d-glucopyranosyl-2,4-dihydroxy-6-oxo-cyclohexa-2,4-dienyl acetic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C14H16O10 | |
| Molar mass | 344.272 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Anthocyanone A is a degradation product of malvidin 3-O-glucoside under acidic conditions.[1] It is found in wine.[2]
References
- ^ Lopes, P; Richard, T; Saucier, C; Teissedre, PL; Monti, JP; Glories, Y (2007). "Anthocyanone A: A quinone methide derivative resulting from malvidin 3-O-glucoside degradation". Journal of Agricultural and Food Chemistry. 55 (7): 2698–704. Bibcode:2007JAFC...55.2698L. doi:10.1021/jf062875o. PMID 17338545.
- ^ Saucier, Cédric (2010). "How do wine polyphenols evolve during wine ageing?". Cerevisia. 35: 11–15. doi:10.1016/j.cervis.2010.05.002.
