1000 Genomes Project
The 1000 Genomes Project (1KGP), taken place from January 2008 to 2015, was an international research effort to establish the most detailed catalogue of human genetic variation at the time. Scientists planned to sequence the genomes of at least one thousand anonymous healthy participants from a number of different ethnic groups within the following three years, using advancements in newly developed technologies. In 2010, the project finished its pilot phase, which was described in detail in a publication in the journal Nature.[1] In 2012, the sequencing of 1092 genomes was announced in a Nature publication.[2] In 2015, two papers in Nature reported results and the completion of the project and opportunities for future research.[3][4]
Many rare variations, restricted to closely related groups, were identified, and eight structural-variation classes were analyzed.[5]
The project united multidisciplinary research teams from institutes around the world, including China, Italy, Japan, Kenya, Nigeria, Peru, the United Kingdom, and the United States contributing to the sequence dataset and to a refined human genome map freely accessible through public databases to the scientific community and the general public alike.[2]
The International Genome Sample Resource was created to host and expand on the data set after the project's end.[6]

Background
Since the completion of the Human Genome Project advances in human population genetics and comparative genomics enabled further insight into genetic diversity.[7] The understanding about structural variations (insertions/deletions (indels), copy number variations (CNV), retroelements), single-nucleotide polymorphisms (SNPs), and natural selection were being solidified.[8][9][10][11]
The diversity of Human genetic variation such as that Indels were being uncovered and investigating human genomic variations[citation needed]
Natural selection
It also aimed to provide evidence that can be used to explore the impact of natural selection on population differences. Patterns of DNA polymorphisms can be used to reliably detect signatures of selection and may help to identify genes that might underlie variation in disease resistance or drug metabolism.[12][13] Such insights could improve understanding of phenotypic variations, genetic disorders and Mendelian inheritance and their effects on survival and/or reproduction of different human populations.
Project description
Goals
The 1000 Genomes Project was designed to bridge the gap of knowledge between rare genetic variants that have a severe effect predominantly on simple traits (e.g. cystic fibrosis, Huntington disease) and common genetic variants have a mild effect and are implicated in complex traits (e.g. cognition, diabetes, heart disease).[14]
The primary goal of this project was to create a complete and detailed catalogue of human genetic variations, which can be used for association studies relating genetic variation to disease. The consortium aimed to discover >95 % of the variants (e.g. SNPs, CNVs, indels) with minor allele frequencies as low as 1% across the genome and 0.1-0.5% in gene regions, as well as to estimate the population frequencies, haplotype backgrounds and linkage disequilibrium patterns of variant alleles.[15]
Secondary goals included the support of better SNP and probe selection for genotyping platforms in future studies and the improvement of the human reference sequence. The completed database was expected be a useful tool for studying regions under selection, variation in multiple populations and understanding the underlying processes of mutation and recombination.[15]
Outline
The human genome consists of approximately 3 billion DNA base pairs and is estimated to carry around 20,000 protein coding genes. In designing the study the consortium needed to address several critical issues regarding the project metrics such as technology challenges, data quality standards and sequence coverage.[15]
Over the course of the next three years,[clarification needed] scientists at the Sanger Institute, BGI Shenzhen and the National Human Genome Research Institute’s Large-Scale Sequencing Network planned to sequence a minimum of 1,000 human genomes. Due to the large amount of sequence data that was required, recruiting additional participants was maintained.[14]
Almost 10 billion bases were to be sequenced per day over a period of the two year production phase, equating to more than two human genomes every 24 hours. The intended sequence dataset was to comprise 6 trillion DNA bases, 60-fold more sequence data than what has been published in DNA databases at the time.[14]
To determine the final design of the full project three pilot studies were to be carried out within the first year of the project. The first pilot intends to genotype 180 people of 3 major geographic groups at low coverage (2×). For the second pilot study, the genomes of two nuclear families (both parents and an adult child) are going to be sequenced with deep coverage (20× per genome). The third pilot study involves sequencing the coding regions (exons) of 1,000 genes in 1,000 people with deep coverage (20×).[14][15]
It was estimated that the project would likely cost more than $500 million if standard DNA sequencing technologies were used. Several newer technologies (e.g. Solexa, 454, SOLiD) were to be applied, lowering the expected costs to between $30 million and $50 million. The major support was provided by the Wellcome Trust Sanger Institute in Hinxton, England; the Beijing Genomics Institute, Shenzhen (BGI Shenzhen), China; and the NHGRI, part of the National Institutes of Health (NIH).[14]
In keeping with Fort Lauderdale principles[16] all genome sequence data (including variant calls) is freely available as the project progresses and can be downloaded via ftp from the 1000 genomes project webpage.[17]
Human genome samples
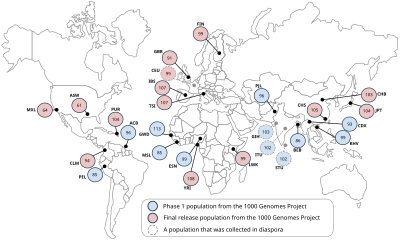
Based on the overall goals for the project, the samples will be chosen to provide power in populations where association studies for common diseases are being carried out. Furthermore, the samples do not need to have medical or phenotype information since the proposed catalogue will be a basic resource on human variation.[15]
For the pilot studies human genome samples from the HapMap collection will be sequenced. It will be useful to focus on samples that have additional data available (such as ENCODE sequence, genome-wide genotypes, fosmid-end sequence, structural variation assays, and gene expression) to be able to compare the results with those from other projects.[15]
Complying with extensive ethical procedures, the 1000 Genomes Project will then use samples from volunteer donors. The following populations will be included in the study: Yoruba in Ibadan (YRI), Nigeria; Japanese in Tokyo (JPT); Chinese in Beijing (CHB); Utah residents with ancestry from northern and western Europe (CEU); Luhya in Webuye, Kenya (LWK); Maasai in Kinyawa, Kenya (MKK); Toscani in Italy (TSI); Peruvians in Lima, Peru (PEL); Gujarati Indians in Houston (GIH); Chinese in metropolitan Denver (CHD); people of Mexican ancestry in Los Angeles (MXL); and people of African ancestry in the southwestern United States (ASW).[14]
* Population that was collected in diaspora
Community meeting
Data generated by the 1000 Genomes Project is widely used by the genetics community, making the first 1000 Genomes Project one of the most cited papers in biology.[19] To support this user community, the project held a community analysis meeting in July 2012 that included talks highlighting key project discoveries, their impact on population genetics and human disease studies, and summaries of other large-scale sequencing studies.[20]
Project findings
Pilot phase
The pilot phase consisted of three projects:
- low-coverage whole-genome sequencing of 179 individuals from 4 populations
- high-coverage sequencing of 2 trios (mother-father-child)
- exon-targeted sequencing of 697 individuals from 7 populations
It was found that on average, each person carries around 250–300 loss-of-function variants in annotated genes and 50-100 variants previously implicated in inherited disorders. Based on the two trios, it is estimated that the rate of de novo germline mutation is approximately 10−8 per base per generation.[1]
See also
- Human Genome Project
- HapMap Project
- Personal genomics
- Population groups in biomedicine
- 1000 Plant Genomes Project
- List of biological databases
References
- ^ a b Abecasis GR, Altshuler D, Auton A, Brooks LD, Durbin RM, Gibbs RA, et al. (October 2010). "A map of human genome variation from population-scale sequencing". Nature. 467 (7319): 1061–73. Bibcode:2010Natur.467.1061T. doi:10.1038/nature09534. PMC 3042601. PMID 20981092.
- ^ a b Abecasis GR, Auton A, Brooks LD, DePristo MA, Durbin RM, Handsaker RE, et al. (November 2012). "An integrated map of genetic variation from 1,092 human genomes". Nature. 491 (7422): 56–65. Bibcode:2012Natur.491...56T. doi:10.1038/nature11632. PMC 3498066. PMID 23128226.
- ^ Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM, Korbel JO, et al. (October 2015). "A global reference for human genetic variation". Nature. 526 (7571): 68–74. Bibcode:2015Natur.526...68T. doi:10.1038/nature15393. PMC 4750478. PMID 26432245.
- ^ Sudmant PH, Rausch T, Gardner EJ, Handsaker RE, Abyzov A, Huddleston J, et al. (October 2015). "An integrated map of structural variation in 2,504 human genomes". Nature. 526 (7571): 75–81. Bibcode:2015Natur.526...75.. doi:10.1038/nature15394. PMC 4617611. PMID 26432246.
- ^ "Variety of life". Nature News & Comment. 2015-09-30. Retrieved 2015-10-15.
- ^ "1000 Genomes Project | Scientific Computing and Data". Mount Sinai School of Medicine. 2020-07-07. Retrieved 2023-10-01.
- ^ Nielsen R (October 2010). "Genomics: In search of rare human variants". Nature. 467 (7319): 1050–1. Bibcode:2010Natur.467.1050N. doi:10.1038/4671050a. PMID 20981085.
- ^ JC Long, Human Genetic Variation: The mechanisms and results of microevolution, American Anthropological Association (2004)
- ^ Anzai T, Shiina T, Kimura N, Yanagiya K, Kohara S, Shigenari A, et al. (June 2003). "Comparative sequencing of human and chimpanzee MHC class I regions unveils insertions/deletions as the major path to genomic divergence". Proceedings of the National Academy of Sciences of the United States of America. 100 (13): 7708–13. Bibcode:2003PNAS..100.7708A. doi:10.1073/pnas.1230533100. PMC 164652. PMID 12799463.
- ^ Redon R, Ishikawa S, Fitch KR, Feuk L, Perry GH, Andrews TD, et al. (November 2006). "Global variation in copy number in the human genome". Nature. 444 (7118): 444–54. Bibcode:2006Natur.444..444R. doi:10.1038/nature05329. PMC 2669898. PMID 17122850.
- ^ Barreiro LB, Laval G, Quach H, Patin E, Quintana-Murci L (March 2008). "Natural selection has driven population differentiation in modern humans". Nature Genetics. 40 (3): 340–5. doi:10.1038/ng.78. PMID 18246066. S2CID 205357396.
- ^ EE Harris et al., The molecular signature of selection underlying human adaptations, Yearbook of Physical Anthropology 49: 89-130 (2006)
- ^ Bamshad M, Wooding SP (February 2003). "Signatures of natural selection in the human genome". Nature Reviews. Genetics. 4 (2): 99–111. doi:10.1038/nrg999. PMID 12560807. S2CID 13722452.
- ^ a b c d e f G Spencer, International Consortium Announces the 1000 Genomes Project, EMBARGOED (2008) http://www.nih.gov/news/health/jan2008/nhgri-22.htm
- ^ a b c d e f Meeting Report: A Workshop to Plan a Deep Catalog of Human Genetic Variation, (2007) http://www.1000genomes.org/sites/1000genomes.org/files/docs/1000Genomes-MeetingReport.pdf
- ^ [https://web.archive.org/web/20131228183230/http://www.genome.gov/pages/research/wellcomereport0303.pdf Archived 2013-12-28 at the Wayback Machine
- ^ 1000 genomes project data webpage
- ^ Oleksyk TK, Brukhin V, O'Brien SJ (2015). "The Genome Russia project: closing the largest remaining omission on the world Genome map". GigaScience. 4: 53. doi:10.1186/s13742-015-0095-0. PMC 4644275. PMID 26568821.
- ^ C. King (2012) The Hottest Research of 2011. Science Watch http://archive.sciencewatch.com/newsletter/2012/201203/hottest_research_2012/
- ^ 1000 Genomes Project Community Analysis Meeting http://1000gconference.sph.umich.edu/
External links
- 1000 Genomes - A Deep Catalog of Human Genetic Variation - official web page
- International HapMap Project Archived 2014-04-16 at the Wayback Machine - official web page
- Human Genome Project Information

