Pitolisant
 | |
| Clinical data | |
|---|---|
| Pronunciation | /pɪˈtɒlɪsənt/ pi-TOL-i-sənt |
| Trade names | Wakix, Ozawade |
| Other names | Tiprolisant; Ciproxidine; BF2.649 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619055 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Histamine H3 receptor inverse agonist |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 10–12 hours |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
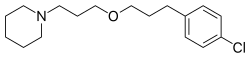
| Formula | C17H26ClNO |
| Molar mass | 295.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Pitolisant, sold under the brand name Wakix among others, is a medication used for the treatment of excessive daytime sleepiness in adults with narcolepsy.[3] It is an inverse agonist of the histamine 3 (H3) receptor (an antihistamine drug specific to that kind of receptors).[3] It represents the first commercially available medication in its class, so that the U.S. Food and Drug Administration (FDA) declares it a first-in-class medication.[7][8] Pitolisant enhances the activity of histaminergic neurons in the brain that function to improve a person's wakefulness.[9] It was approved by the European Medicines Agency (EMA) in March 2016 for narcolepsy with or without cataplexy, and for excessive daytime sleepiness by the FDA in August 2019.[10] The most common side effects include difficulty sleeping, nausea, and feeling worried.[11]
Medical uses
Pitolisant is indicated in adults for the treatment of narcolepsy.[3][4] Narcolepsy is a chronic sleep disorder that causes overwhelming daytime drowsiness.[4] Pitolisant is also indicated to improve alertness and reduce excessive daytime sleepiness in adults with obstructive sleep apnea.[5][12]
Side effects
The most common side effects include insomnia, headache, nausea, anxiety, irritability, dizziness, depression, tremor, sleep disorders, tiredness, vomiting, vertigo, dyspepsia, and heartburn.[4] Rare but serious side effects are abnormal weight loss and spontaneous abortion.[4]
Pharmacology
Pitolisant is an inverse agonist of the histamine 3 (H3) autoreceptor. The H3 autoreceptors regulate histaminergic activity in the central nervous system (and to a lesser extent, the peripheral nervous system) by inhibiting histamine synthesis and release upon binding to endogenous histamine.[13] By preventing the binding of endogenous histamine at the H3, as well as producing a response opposite to that of endogenous histamine at the receptor (inverse agonism), pitolisant enhances histaminergic activity in the brain.[14]
Pitolisant is a drug that belongs to the class of CNS stimulants.[15][16][17][18] Pitolisant is also considered a medication of class "eugeroic", which means that it promotes wakefulness and alertness. Eugeroics are different from traditional CNS stimulants such as amphetamine in that they have fewer side effects and lower abuse potential. Pitolisant is the first eugeroic drug that acts by blocking the histamine 3 (H3) autoreceptor, which increases the activity of histamine neurons in the brain. Pitolisant has been shown to be effective and well-tolerated for the treatment of narcolepsy with or without cataplexy.[19][20][21]
| Target | Ki | Activity |
|---|---|---|
| Histamine-3 | 150 nM | Inverse agonist |
| Sigma-1 | <10 nM | Antagonist |
| Sigma-2 | 52 nM | Antagonist |
| D3 | 382 nM | Antagonist |
| 5HT2A | 544 nM | Antagonist |
Pitolisant has been demonstrated to exhibit high affinity for sigma-1 and sigma-2 receptors, as well as moderate affinity for 5HT2A and D3 receptors. There exist conflicting findings relating the intrinsic activity of pitolisant at the 5HT2A receptor.[23]
Pharmacokinetics
Pitolisant is readily absorbed when taken by mouth and reaches peak blood concentrations approximately 3 hours after administration. The biological half-life of Pitolisant ranges from 10 to 12 hours.[14]
History
Pitolisant is marketed in the European Union by Bioprojet Pharma.[4] It was approved for medical use in the European Union in March 2016 by the European Medicines Agency (EMA).[10][4]
The U.S. Food and Drug Administration (FDA) approved pitolisant for excessive daytime sleepiness in participants with narcolepsy based primarily on evidence from two trials (Trial 1/NCT01067222, Trial 2/NCT01638403).[11] An additional trial (Trial 3/NCT01800045), in which participants with a different type of narcolepsy were exposed to the same dose of pitolisant, was used to add data for evaluation of side effects.[11] The trials were conducted in Europe and South America.[11]
The two primary trials enrolled adults with narcolepsy and excessive daytime sleepiness.[11] Participants received pitolisant, placebo, or an approved drug for narcolepsy for eight weeks.[11] For participants receiving pitolisant, the dose could be increased during the first three weeks but had to remain the same for the next five weeks.[11] Neither the participants nor the healthcare providers knew which treatment was being given during the trial.[11]
The benefit of pitolisant was evaluated by comparing changes in daytime sleepiness during the trial between pitolisant- and placebo-treated participants.[11] To measure the daytime sleepiness, the investigators used a scale called the Epworth Sleepiness Scale (ESS).[11] The ESS asks participants to rate the likelihood that they would fall asleep while doing eight daily activities (such as sitting and reading or watching television).[11] Participants rate each item from zero (would never doze) to three (high chance of dozing).[11]
Pitolisant was approved by the FDA in August 2019.[10][11] It was granted orphan drug designation for the treatment of narcolepsy,[24] fast track designation for the treatment of excessive daytime sleepiness and cataplexy in people with narcolepsy, and breakthrough therapy designation for the treatment of cataplexy in people with narcolepsy.[25]
Society and culture
Legal status
Pitolisant is approved in the European Union and the United States to treat narcolepsy, and is not a controlled substance in these countries.[failed verification] Still, long-term studies comparing the effectiveness and tolerability of pitolisant with modafinil or sodium oxybate are lacking.[failed verification] Pitolisant, the only non-controlled anti-narcoleptic drug in the US,[21] has shown minimal abuse risk in studies.[21][26]
References
- ^ "Summary Basis of Decision (SBD) for Wakix". Health Canada. 23 October 2014. Archived from the original on 22 December 2022. Retrieved 29 May 2022.
- ^ "Health product highlights 2021: Annexes of products approved in 2021". Health Canada. 3 August 2022. Archived from the original on 25 March 2024. Retrieved 25 March 2024.
- ^ a b c d "Wakix- pitolisant hydrochloride tablet, film coated". DailyMed. 6 November 2019. Archived from the original on 11 August 2020. Retrieved 18 August 2020.
- ^ a b c d e f g "Wakix EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 12 November 2020. Retrieved 18 August 2020. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b "Ozawade EPAR". European Medicines Agency (EMA). 20 May 2021. Archived from the original on 15 October 2021. Retrieved 15 October 2021.
- ^ "Ozawade Product information". Union Register of medicinal products. Archived from the original on 5 March 2023. Retrieved 3 March 2023.
- ^ "New Drug Therapy Approvals 2019". U.S. Food and Drug Administration (FDA). 31 December 2019. Archived from the original on 16 September 2020. Retrieved 15 September 2020.
- ^ "FDA Approves Pitolisant for Daytime Sleepiness in Patients with Narcolepsy". Pharmacy Times. 16 August 2019. Archived from the original on 12 August 2020. Retrieved 18 August 2020.
- ^ Syed YY (September 2016). "Pitolisant: First Global Approval". Drugs. 76 (13): 1313–1318. doi:10.1007/s40265-016-0620-1. PMID 27438291. S2CID 42684839.
- ^ a b c Giliberto S, Shishodia R, Nastruz M, Brar C, Bulathsinhala S, Terry J, Pemminati S, Shenoy SK (March 2024). "A Comprehensive Review of Novel FDA-Approved Psychiatric Medications (2018-2022)". Cureus. 16 (3): e56561. doi:10.7759/cureus.56561. PMC 11028406. PMID 38646400.
- ^ a b c d e f g h i j k l m "Drug Trials Snapshots: Wakix". U.S. Food and Drug Administration (FDA). 14 August 2019. Archived from the original on 22 December 2020. Retrieved 18 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Neshat SS, Heidari A, Henriquez-Beltran M, Patel K, Colaco B, Arunthari V, Lee Mateus AY, Cheung J, Labarca G (August 2024). "Evaluating pharmacological treatments for excessive daytime sleepiness in obstructive sleep apnea: A comprehensive network meta-analysis and systematic review". Sleep Med Rev. 76: 101934. doi:10.1016/j.smrv.2024.101934. PMID 38754208.
- ^ West RE, Zweig A, Shih NY, Siegel MI, Egan RW, Clark MA (November 1990). "Identification of two H3-histamine receptor subtypes". Molecular Pharmacology. 38 (5): 610–613. PMID 2172771. Archived from the original on 10 December 2023. Retrieved 10 April 2023.
- ^ a b Sarfraz N, Okuampa D, Hansen H, Alvarez M, Cornett EM, Kakazu J, et al. (30 May 2022). "pitolisant, a novel histamine-3 receptor competitive antagonist, and inverse agonist, in the treatment of excessive daytime sleepiness in adult patients with narcolepsy". Health Psychology Research. 10 (3): 34222. doi:10.52965/001c.34222. PMC 9239364. PMID 35774905.
- ^ "Pitolisant". National Institute of Diabetes and Digestive and Kidney Diseases. 2012. PMID 34516055. Archived from the original on 14 November 2023. Retrieved 22 January 2024.
- ^ "Central Nervous System (CNS) Stimulants". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 22 January 2012. PMID 31644012. Archived from the original on 29 January 2023. Retrieved 22 January 2024 – via PubMed.
- ^ "Pitolisant Uses, Side Effects & Warnings". Drugs.com. Archived from the original on 19 January 2024. Retrieved 22 January 2024.
- ^ "List of CNS stimulants + Uses & Side Effects". Drugs.com. Archived from the original on 6 January 2024. Retrieved 22 January 2024.
- ^ "Pitolisant (Wakix) for Narcolepsy". JAMA. 326 (11): 1060–1061. 21 September 2021. doi:10.1001/jama.2021.1349. PMID 34546302. S2CID 237583921. Archived from the original on 8 March 2024. Retrieved 22 January 2024 – via Silverchair.
- ^ Kollb-Sielecka M, Demolis P, Emmerich J, Markey G, Salmonson T, Haas M (1 May 2017). "The European Medicines Agency review of pitolisant for treatment of narcolepsy: summary of the scientific assessment by the Committee for Medicinal Products for Human Use". Sleep Medicine. 33: 125–129. doi:10.1016/j.sleep.2017.01.002. PMID 28449891. Archived from the original on 8 March 2024. Retrieved 22 January 2024 – via Europe PMC.
- ^ a b c Lamb YN (February 2020). "Pitolisant: A Review in Narcolepsy with or without Cataplexy". CNS Drugs. 34 (2): 207–218. doi:10.1007/s40263-020-00703-x. PMID 31997137. S2CID 210949049.
- ^ Shah D (12 July 2019). "OPDP Labeling Comments for WAKIX® (pitolisant) tablets, for oral use" (PDF). www.accessdata.fda.gov. Archived (PDF) from the original on 30 September 2024. Retrieved 28 September 2024.
- ^ Solages M. "Wakix (Pitolisant)" (PDF). www.accessdata.fda.gov. Archived (PDF) from the original on 28 September 2024. Retrieved 28 September 2024.
- ^ "Pitolisant Orphan Drug Designations and Approvals". U.S. Food and Drug Administration (FDA). 17 May 2010. Archived from the original on 26 May 2021. Retrieved 25 May 2021.
- ^ "Harmony's pitolisant granted breakthrough and fast track designations". Pharma Business International. 22 May 2018. Archived from the original on 26 May 2021. Retrieved 25 May 2021.
- ^ de Biase S, Pellitteri G, Gigli GL, Valente M (February 2021). "Evaluating pitolisant as a narcolepsy treatment option". Expert Opinion on Pharmacotherapy. 22 (2): 155–162. doi:10.1080/14656566.2020.1817387. PMID 32941089. S2CID 221788777.
External links
- Clinical trial number NCT01067222 for "Efficacy and Safety Study of BF2.649 in the Treatment of Excessive Daytime Sleepiness in Narcolepsy (Harmony1)" at ClinicalTrials.gov
- Clinical trial number NCT01638403 for "Effects of BF2.649 in the Treatment of Excessive Daytime Sleepiness in Narcolepsy." at ClinicalTrials.gov
