Lutetium compounds
Lutetium compounds are compounds formed by the lanthanide metal lutetium (Lu). In these compounds, lutetium generally exhibits the +3 oxidation state, such as LuCl3, Lu2O3 and Lu2(SO4)3.[1] Aqueous solutions of most lutetium salts are colorless and form white crystalline solids upon drying, with the common exception of the iodide. The soluble salts, such as nitrate, sulfate and acetate form hydrates upon crystallization. The oxide, hydroxide, fluoride, carbonate, phosphate and oxalate are insoluble in water.[2]
Oxides

Lutetium(III) oxide is a white solid, a cubic compound of lutetium which sometimes used in the preparation of specialty glasses. It is also called lutecia. It is a lanthanide oxide, also known as a rare earth.[3][4][5] Lutetium(III) oxide is an important raw material for laser crystals.[6] It also has specialized uses in ceramics, glass, phosphors, and lasers. Lutetium(III) oxide is used as a catalyst in cracking, alkylation, hydrogenation, and polymerization.[3] The band gap of lutetium oxide is 5.5 eV.[7]
Halides
Lutetium(III) fluoride can be produced by reacting lutetium oxide with hydrogen fluoride, or reacting lutetium chloride and hydrofluoric acid.[8] It can also be produced by reacting lutetium sulfide and hydrofluoric acid:[9]
- 3 Lu
2S
3+ 20 HF + (2 + 2x) H
2O → 2 (H
3O)Lu
3F
10·xH
2O↓ + 9 H
2S↑ (x = 0.9) - (H3O)Lu3F10 → 3 LuF3 + HF↑ + H2O↑
Lutetium oxide and nitrogen trifluoride react at 240 °C to produce LuOF. A second step happens below 460 °C to produce LuF3.[10] Lutetium(III) chloride forms hygroscopic white monoclinic crystals[11] and also a hydroscopic hexahydrate LuCl3·6H2O.[12] Anhydrous lutetium(III) chloride has the YCl3 (AlCl3) layer structure with octahedral lutetium ions.[13] Lutetium(III) bromide can be synthesized through the following reaction:[14]
- 2 Lu(s) + 3 Br2(g) → 2 LuBr3(s)
If burned, lutetium(III) bromide may produce hydrogen bromide and metal oxide fumes.[15] Lutetium(III) bromide reacts to strong oxidizing agents.[15] Lutetium(III) iodide can be obtained by reacting lutetium with iodine:[16][17]
- 2 Lu + 3 I2 → LuI3
Lutetium(III) iodide can also obtained by the reacting metallic lutetium with mercury iodide in vacuum at 500 °C:[16]
- 2 Lu + 3 HgI2 → 2 LuI3 + 3 Hg
The elemental mercury generated in the reaction can be removed by distillation.[18] The lutetium(III) iodide hydrate crystallized from the solution can be heated with ammonium iodide to obtain the anhydrate.[19][16]
Coordination compounds
Nitrogen-containing ligand complexes
Lutetium phtalocyanine

Lutetium phthalocyanine is the most notable coordination compound of lutetium, and is derived from lutetium and two phthalocyanines. It was the first known example of a molecule that is an intrinsic semiconductor.[20][21] It exhibits electrochromism, changing color when subject to a voltage. It is a double-decker sandwich compound consisting of a Lu3+ ion coordinated to two the conjugate base of two phthalocyanines. The rings are arranged in a staggered conformation. The extremities of the two ligands are slightly distorted outwards.[22] The complex features a non-innocent ligand, in the sense that the macrocycles carry an extra electron.[23] It is a free radical[20] with the unpaired electron sitting in a half-filled molecular orbital between the highest occupied and lowest unoccupied orbitals, allowing its electronic properties to be finely tuned.[22] It, along with many substituted derivatives like the alkoxy-methyl derivative Lu[(C8H17OCH2)8Pc]2, can be deposited as a thin film with intrinsic semiconductor properties;[23] said properties arise due to its radical nature[20] and its low reduction potential compared to other metal phthalocyanines.[21] This initially green film exhibits electrochromism; the oxidized form LuPc+
2 is red, whereas the reduced form LuPc−
2 is blue and the next two reduced forms are dark blue and violet, respectively.[23] The green/red oxidation cycle can be repeated over 10,000 times in aqueous solution with dissolved alkali metal halides, before it is degraded by hydroxide ions; the green/blue redox degrades faster in water.[23]
Other complexes
[LuI2(HOiPr)4]I can be dissolved in pyridine-THF to give yellow [LuI(OiPr)(py)5]I. LuI3 is directly dissolved in pyridine-THF to obtain yellow [LuI2(py)5]I. In both compounds pyridine is coordinated to lutetium by nitrogen atom.[24] Lutetium(III) nitrate can be crystallized with 2,2':6',2-terpyridine (terpy) in dry acetonitrile to obtain colorless [Lu(terpy)(NO3)3], in which the nitrogen atom and the oxygen atom of the nitrate are coordinated to the lutetium atom.[25]
Oxygen-containing ligand complexes
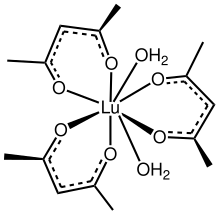
Trivalent lutetium and water can form complex ions such as [Lu(OH2)n]3+, and lutetium(III) perchlorate and lutetium(III) trifluoromethanesulfonate can exist in the form of hydrates.[26] Ether (R2O) is also a common oxygen-containing ligand. For example, Lu(CH2SiMe3)3(THF)2 can be obtained by reacting lutetium(III) chloride and (trimethylsilyl)methyllithium in a solvent containing tetrahydrofuran (THF).[27]
Other compounds
Adding ammonia water or a hydroxide to the aqueous solution of any soluble lutetium salt can precipitate lutetium(III) hydroxide (Lu(OH)3). The hexagonal lutetium hydroxide can be heated and dehydrated to obtain the monoclinic lutetium oxyhydroxide (LuO(OH)), and further heating will make it decompose into lutetium(III) oxide (Lu2O3).[28] Lutetium oxyhalides (LuOX, X=Cl, Br, I) can be obtained by hydrolysis of the lutetium trihalides.[28] Lu2Cl2C can be obtained by reacting lutetium(III) chloride, caesium chloride, lutetium and carbon at a high temperature.[29]
References
- ^ "Lutetium".
- ^ Patnaik, Pradyot (2003). Handbook of Inorganic Chemical Compounds. McGraw-Hill. p. 510. ISBN 978-0-07-049439-8. Retrieved 2009-06-06.
- ^ a b Lutetium Oxide. 1997-2007. Metall Rare Earth Limited. http://www.metall.com.cn/luo.htm
- ^ Macintyre, J. E. (1992). Dictionary of Inorganic Compounds Volumes 1–3. London: Chapman & Hall.
- ^ Trotman-Dickenson, A. F. (1973). Comprehensive Inorganic Chemistry. Oxford: Pergamon.
- ^ Parsonage, Tina L.; Beecher, Stephen J.; Choudhary, Amol; Grant-Jacob, James A.; Hua, Ping; MacKenzie, Jacob I.; Shepherd, David P.; Eason, Robert W. (2015). "Pulsed laser deposited diode-pumped 7.4 W Yb:Lu2O3 planar waveguide laser" (PDF). Optics Express. 23 (25): 31691–7. Bibcode:2015OExpr..2331691P. doi:10.1364/oe.23.031691. PMID 26698962.
- ^ Ordin, S. V.; Shelykh, A. I. (2010). "Optical and dielectric characteristics of the rare-earth metal oxide Lu2O3". Semiconductors. 44 (5): 558–563. Bibcode:2010Semic..44..558O. doi:10.1134/S1063782610050027. S2CID 101643906.
- ^ Georg Brauer (ed.), In collaboration with Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 254.
- ^ O.V. Andrrev, I.A. Razumkova, A.N. Boiko (March 2018). "Synthesis and thermal stability of rare earth compounds REF 3 , REF 3 · n H 2 O and (H 3 O)RE 3 F 10 · n H 2 O (RE = Tb − Lu, Y), obtained from sulphide precursors". Journal of Fluorine Chemistry. 207: 77–83. doi:10.1016/j.jfluchem.2017.12.001.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Randall D. Scheele, Bruce K. McNamara, Andrew M. Casella, Anne E. Kozelisky, Doinita Neiner (February 2013). "Thermal NF3 fluorination/oxidation of cobalt, yttrium, zirconium, and selected lanthanide oxides". Journal of Fluorine Chemistry. 146: 86–97. doi:10.1016/j.jfluchem.2012.12.013.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, p. 472, ISBN 0-8493-0594-2, retrieved 2008-06-27
- ^ "Lutetium(III) chloride hexahydrate 542075". Sigma-Aldrich. Retrieved 2019-07-24.
- ^ Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- ^ Winter, Mark. "Lutetium»reactions of elements [WebElements Periodic Table]". www.webelements.com. Retrieved 22 December 2016.
- ^ a b "Lutetian bromide" (PDF). SDS. Retrieved 22 December 2016.
- ^ a b c Georg Brauer (Hrsg.), unter Mitarbeit von Marianne Baudler u. a.: Handbuch der Präparativen Anorganischen Chemie. 3., umgearbeitete Auflage. Band I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6, S. 1077.
- ^ Webelements: Lutetium: lutetium triiodide Retrieved 31.3.2018
- ^ Asprey, L. B.; Keenan, T. K.; Kruse, F. H. Preparation and crystal data for lanthanide and actinide triiodides. Inorg. Chem., 1964. 3 (8): 1137-1240
- ^ 无机化学丛书 第七卷 钪 稀土元素. 科学出版社. pp 211
- ^ a b c Belarbi, Z.; Sirlin, C.; Simon, J.; Andre, Jean Jacques (November 1989). "Electrical and magnetic properties of liquid crystalline molecular materials: lithium and lutetium phthalocyanine derivatives". The Journal of Physical Chemistry. 93 (24): 8105–8110. doi:10.1021/j100361a026.
- ^ a b Trometer, M.; Even, R.; Simon, J.; Dubon, A.; Laval, J.-Y.; Germain, J.P.; Maleysson, C.; Pauly, A.; Robert, H. (May 1992). "Lutetium bisphthalocyanine thin films for gas detection". Sensors and Actuators B: Chemical. 8 (2): 129–135. doi:10.1016/0925-4005(92)80169-X.
- ^ a b Bidermane, I.; Lüder, J.; Boudet, S.; Zhang, T.; Ahmadi, S.; Grazioli, C.; Bouvet, M.; Rusz, J.; Sanyal, B.; Eriksson, O.; Brena, B.; Puglia, C.; Witkowski, N. (21 June 2013). "Experimental and theoretical study of electronic structure of lutetium bi-phthalocyanine". The Journal of Chemical Physics. 138 (23): 234701. Bibcode:2013JChPh.138w4701B. doi:10.1063/1.4809725. ISSN 0021-9606. PMID 23802970.
- ^ a b c d Toupance, Thierry; Plichon, Vincent; Simon, Jacques (1999). "Substituted bis(phthalocyanines): electrochemical properties and probe beam deflection (mirage) studies". New Journal of Chemistry. 23 (10): 1001–1006. doi:10.1039/A905248H.
- ^ Garth R. Giesbrecht, John C. Gordon, David L. Clark, Brian L. Scott (2004-02-01). "Auto-ionization in Lutetium Iodide Complexes: Effect of the Ionic Radius on Lanthanide−Iodide Binding". Inorganic Chemistry. 43 (3): 1065–1070. doi:10.1021/ic035090y. ISSN 0020-1669. PMID 14753829. Retrieved 2022-12-29.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Birte Ahrens, Simon A. Cotton, Neil Feeder, Oliver E. Noy, Paul R. Raithby, Simon J. Teat (2002-04-26). "Structural variety in nitrate complexes of the heavy lanthanides with 2,2′:6′,2″-terpyridine, and stereoselective replacement of nitrate". Journal of the Chemical Society, Dalton Transactions (9): 2027–2030. doi:10.1039/b200480c. Retrieved 2022-12-29.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Simon A. Cotton, Paul R. Raithby, Alexander Shield, Jack M. Harrowfield (March 2022). "A comparison of the structural chemistry of scandium, yttrium, lanthanum and lutetium: A contribution to the group 3 debate". Coordination Chemistry Reviews. 455: 214366. doi:10.1016/j.ccr.2021.214366. S2CID 245712597. Retrieved 2022-12-29.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Konstantin A. Rufanov, Dominique M. M. Freckmann, Heinz-Jürgen Kroth, Stefan Schutte, Herbert Schumann (2005-05-01). "Studies on the Thermolysis of Ether-Stabilized Lu(CH 2 SiMe 3 ) 3 . Molecular Structure of Lu(CH 2 SiMe 3 ) 3 (THF)(diglyme)". Zeitschrift für Naturforschung B. 60 (5): 533–537. doi:10.1515/znb-2005-0509. ISSN 1865-7117. S2CID 100903579.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b 无机化学丛书. pp 206. 1.3.3 氧化态+3的化合物.
- ^ Thomas Schleid, Gerd Meyer (September 1987). "Synthesis and crystal structures of hydrogen and carbon stabilized lutetium monochloride, LuClHx and Lu2Cl2C". Zeitschrift für anorganische und allgemeine Chemie (in German). 552 (9): 90–96. doi:10.1002/zaac.19875520909. ISSN 0044-2313. Retrieved 2022-12-29.
