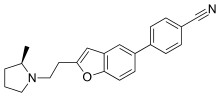
ABT-239
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C22H22N2O |
| Molar mass | 330.431 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
ABT-239 is an H3-receptor inverse agonist developed by Abbott. It has stimulant and nootropic effects, and has been investigated as a treatment for ADHD, Alzheimer's disease, and schizophrenia.[1][2][3][4] ABT-239 is more active at the human H3 receptor than comparable agents such as thioperamide, ciproxifan, and cipralisant. It was ultimately dropped from human trials after showing the dangerous cardiac side effect of QT prolongation,[5] but is still widely used in animal research into H3 antagonists / inverse agonists.
References
- ^ Esbenshade TA, Fox GB, Krueger KM, Miller TR, Kang CH, Denny LI, Witte DG, Yao BB, Pan L, Wetter J, Marsh K, Bennani YL, Cowart MD, Sullivan JP, Hancock AA (2005). "Pharmacological properties of ABT-239 [4-(2-{2-[(2R)-2-Methylpyrrolidinyl]ethyl}-benzofuran-5-yl)benzonitrile]: I. Potent and selective histamine H3 receptor antagonist with drug-like properties". J. Pharmacol. Exp. Ther. 313 (1): 165–75. doi:10.1124/jpet.104.078303. PMID 15608078. S2CID 573355.
- ^ Fox GB, Esbenshade TA, Pan JB, Radek RJ, Krueger KM, Yao BB, Browman KE, Buckley MJ, Ballard ME, Komater VA, Miner H, Zhang M, Faghih R, Rueter LE, Bitner RS, Drescher KU, Wetter J, Marsh K, Lemaire M, Porsolt RD, Bennani YL, Sullivan JP, Cowart MD, Decker MW, Hancock AA (2005). "Pharmacological properties of ABT-239 [4-(2-{2-[(2R)-2-Methylpyrrolidinyl]ethyl}-benzofuran-5-yl)benzonitrile]: II. Neurophysiological characterization and broad preclinical efficacy in cognition and schizophrenia of a potent and selective histamine H3 receptor antagonist". J. Pharmacol. Exp. Ther. 313 (1): 176–90. doi:10.1124/jpet.104.078402. PMID 15608077. S2CID 15430117.
- ^ Cowart M, Faghih R, Curtis MP, Gfesser GA, Bennani YL, Black LA, Pan L, Marsh KC, Sullivan JP, Esbenshade TA, Fox GB, Hancock AA (2005). "4-(2-[2-(2(R)-methylpyrrolidin-1-yl)ethyl]benzofuran-5-yl)benzonitrile and related 2-aminoethylbenzofuran H3 receptor antagonists potently enhance cognition and attention". J. Med. Chem. 48 (1): 38–55. doi:10.1021/jm040118g. PMID 15634000.
- ^ Le S, Gruner JA, Mathiasen JR, Marino MJ, Schaffhauser H (June 2008). "Correlation between ex vivo receptor occupancy and wake-promoting activity of selective H3 receptor antagonists". J. Pharmacol. Exp. Ther. 325 (3): 902–9. doi:10.1124/jpet.107.135343. PMID 18305012. S2CID 26536000.
- ^ Hancock, AA (2006). "The challenge of drug discovery of a GPCR target: analysis of preclinical pharmacology of histamine H3 antagonists/inverse agonists". Biochemical Pharmacology. 71 (8): 1103–13. doi:10.1016/j.bcp.2005.10.033. PMID 16513092.
External links
- 4-(2-(2-(2-methyl-1-pyrrolidinyl)ethyl)-1-benzofuran-5-yl)benzonitrile at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
