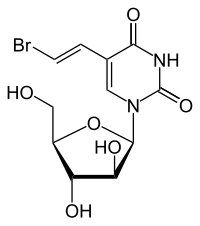
Sorivudine
 | |
| Clinical data | |
|---|---|
| Trade names | Usevir, Brovavir |
| Other names | BV-araU, Bromovinyl araU, 5-Bromovinyl-araU, 5-[(E)-2-bromoethenyl]-1-[(2R,3S,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidine-2,4-dione |
| Routes of administration | Oral |
| ATC code |
|
| Pharmacokinetic data | |
| Metabolism | Viral thymidine kinase |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C11H13BrN2O6 |
| Molar mass | 349.137 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Sorivudine (INN), is a nucleoside analogue antiviral drug, marketed under trade names such as Usevir (Nippon Shoji, Eisai) and Brovavir (BMS). It is used for the treatment of varicella zoster virus infections.[1]
Pharmacology
Feature
- First-line[citation needed] treatment of herpes drug acyclovir was (Zovirax, Activir) from VZV strong activity of the virus.
- Undergoes gastrointestinal absorption, absorption from the gastrointestinal tract after the most degrading without being excreted in urine.
Mechanism of action
- Sorivudine is phosphorylated by thymidine kinase activity in the body and is absorbed into the virus's DNA instead of the correct nucleoside. It is a competitive inhibitor of DNA polymerase, so the viral DNA cannot be replicated and the virus cannot replicate.
Microbiology
Sorivudine is active against most species in the herpesvirus family.
- Herpes simplex virus type I (HSV-1)
- Varicella zoster virus (VZV)
- Epstein–Barr virus (EBV)
Interactions

Sorivudine interacts strongly and in some cases lethally with fluorouracil (5-FU), its prodrugs and related substances. This is based on the metabolite bromovinyluracil (BVU), which irreversibly inhibits the enzyme dihydropyrimidine dehydrogenase (DPD) which is necessary for inactivating 5-FU. The closely related drug brivudine has the same interaction.[2]
Also, it should be taken into consideration that the ability to metabolize this drug can decrease with age due to the composition of the gut microbiota. Specifically, after the age of 60, it has been observed a reduction of the metabolic potential to degrade this compound decreases. [3]
References
- ^ Whitley RJ (1996). "Sorivudine: A Potent Inhibitor of Varicella Zoster Virus Replication". Antiviral Chemotherapy 4. Advances in Experimental Medicine and Biology. Vol. 394. pp. 41–4. doi:10.1007/978-1-4757-9209-6_5. ISBN 978-1-4757-9211-9. PMID 8815706.
- ^ "UAW – Aus Fehlern lernen - Potenziell tödlich verlaufende Wechselwirkung zwischen Brivudin (Zostex) und 5-Fluoropyrimidinen" (PDF). Deutsches Ärzteblatt (in German). 103 (27). 7 July 2006.
- ^ Heinken A, Hertel J, Acharya G, et al. (19 January 2023). "Genome-scale metabolic reconstruction of 7,302 human microorganisms for personalized medicine". Nature Biotechnology. 41 (9): 1320–1331. doi:10.1038/s41587-022-01628-0. PMC 10497413.
