Ilmenite
| Ilmenite | |
|---|---|
 Ilmenite from Miass, Ilmen Mts, Chelyabinsk Oblast', Southern Urals, Urals Region, Russia. 4.5 x 4.3 x 1.5 cm | |
| General | |
| Category | Oxide mineral |
| Formula (repeating unit) | Iron titanium oxide, FeTiO 3 |
| IMA symbol | Ilm[1] |
| Strunz classification | 4.CB.05 |
| Dana classification | 04.03.05.01 |
| Crystal system | Trigonal |
| Crystal class | Rhombohedral (3) H-M symbol: (3) |
| Space group | R3 (no. 148) |
| Unit cell | a = 5.08854(7) c = 14.0924(3) [Å]: Z = 6 |
| Identification | |
| Color | Iron-black; gray with a brownish tint in reflected light |
| Crystal habit | Granular to massive and lamellar exsolutions in hematite or magnetite |
| Twinning | {0001} simple, {1011} lamellar |
| Cleavage | Absent; parting on {0001} and {1011} |
| Fracture | Conchoidal to subconchoidal |
| Tenacity | Brittle |
| Mohs scale hardness | 5–6 |
| Luster | Metallic to submetallic |
| Streak | Black |
| Diaphaneity | Opaque |
| Specific gravity | 4.70–4.79 |
| Optical properties | Uniaxial (–) |
| Birefringence | Strong; O: pinkish brown, E: dark brown (bireflectance) |
| Other characteristics | Weakly magnetic |
| References | [2][3][4] |
Ilmenite is a titanium-iron oxide mineral with the idealized formula FeTiO
3. It is a weakly magnetic black or steel-gray solid. Ilmenite is the most important ore of titanium[5] and the main source of titanium dioxide, which is used in paints, printing inks,[6] fabrics, plastics, paper, sunscreen, food and cosmetics.[7]
Structure and properties
Ilmenite is a heavy (specific gravity 4.7), moderately hard (Mohs hardness 5.6 to 6), opaque black mineral with a submetallic luster.[8] It is almost always massive, with thick tabular crystals being quite rare. It shows no discernible cleavage, breaking instead with a conchoidal to uneven fracture.[9]
Ilmenite crystallizes in the trigonal system with space group R3.[10][3] The ilmenite crystal structure consists of an ordered derivative of the corundum structure; in corundum all cations are identical but in ilmenite Fe2+ and Ti4+ ions occupy alternating layers perpendicular to the trigonal c axis.
Pure ilmenite is paramagnetic (showing only very weak attraction to a magnet), but ilmenite forms solid solutions with hematite that are weakly ferromagnetic and so are noticeably attracted to a magnet. Natural deposits of ilmenite usually contain intergrown or exsolved magnetite that also contribute to its ferromagnetism.[8]
Ilmenite is distinguished from hematite by its less intensely black color and duller appearance and its black streak, and from magnetite by its weaker magnetism.[9][8]
- Crystal structure of ilmenite
- Ilmenite from Froland, Aust-Agder, Norway; 4.1 × 4.1 × 3.8 cm
- Ilmenite and hematite under normal light
- Ilmenite and hematite under polarized light
Discovery
In 1791 William Gregor discovered a deposit of black sand in a stream that runs through the valley just south of the village of Manaccan (Cornwall), and identified for the first time titanium as one of the constituents of the main mineral in the sand.[11][12][13] Gregor named this mineral manaccanite.[14] The same mineral was found in the Ilmensky Mountains, near Miass, Russia, and named ilmenite.[9]
Mineral chemistry
Pure ilmenite has the composition FeTiO3. However, ilmenite most often contains appreciable quantities of magnesium and manganese and up to 6 wt% of hematite, Fe2O3, substituting for FeTiO3 in the crystal structure. Thus the full chemical formula can be expressed as (Fe,Mg,Mn,Ti)O3.[8] Ilmenite forms a solid solution with geikielite (MgTiO
3) and pyrophanite (MnTiO
3) which are magnesian and manganiferous end-members of the solid solution series.[3]
Although ilmenite is typically close to the ideal FeTiO
3 composition, with minor mole percentages of Mn and Mg,[3] the ilmenites of kimberlites usually contain substantial amounts of geikielite molecules,[15] and in some highly differentiated felsic rocks ilmenites may contain significant amounts of pyrophanite molecules.[16]
At temperatures above 950 °C (1,740 °F), there is a complete solid solution between ilmenite and hematite. There is a miscibility gap at lower temperatures, resulting in a coexistence of these two minerals in rocks but no solid solution.[8] This coexistence may result in exsolution lamellae in cooled ilmenites with more iron in the system than can be homogeneously accommodated in the crystal lattice.[17] Ilmenite containing 6 to 13 percent Fe2O3 is sometimes described as ferrian ilmenite.[18][19]
Ilmenite alters or weathers to form the pseudo-mineral leucoxene, a fine-grained yellowish to grayish or brownish material[8][20] enriched to 70% or more of TiO2.[19] Leucoxene is an important source of titanium in heavy mineral sands ore deposits.[21]
Paragenesis
Ilmenite is a common accessory mineral found in metamorphic and igneous rocks.[3] It is found in large concentrations in layered intrusions where it forms as part of a cumulate layer within the intrusion. Ilmenite generally occurs in these cumulates together with orthopyroxene[22] or in combination with plagioclase and apatite (nelsonite).[23]
Magnesian ilmenite is formed in kimberlites as part of the MARID association of minerals (mica-amphibole-rutile-ilmenite-diopside) assemblage of glimmerite xenoliths.[24] Manganiferous ilmenite is found in granitic rocks[16] and also in carbonatite intrusions where it may also contain anomalously high amounts of niobium.[25]
Many mafic igneous rocks contain grains of intergrown magnetite and ilmenite, formed by the oxidation of ulvospinel.[18]
Processing and consumption

Most ilmenite is mined for titanium dioxide production.[26] Ilmenite and titanium dioxide are used in the production of titanium metal.[27][28]
Titanium dioxide is most used as a white pigment and the major consuming industries for TiO2 pigments are paints and surface coatings, plastics, and paper and paperboard. Per capita consumption of TiO2 in China is about 1.1 kilograms per year, compared with 2.7 kilograms for Western Europe and the United States.[29]

Titanium is the ninth most abundant element on Earth and represents about 0.6 percent of the Earth's crust. Ilmenite is commonly processed to obtain a titanium concentrate, which is called "synthetic rutile" if it contains more than 90 percent TiO2, or more generally "titaniferous slags" if it has a lower TiO2 content. More than 80 percent of the estimated global production of titanium concentrate is obtained from the processing of ilmenite, while 13 percent is obtained from titaniferous slags and 5 percent from rutile.[30]
Ilmenite can be converted into pigment grade titanium dioxide via either the sulfate process or the chloride process.[31] Ilmenite can also be improved and purified to titanium dioxide in the form of rutile using the Becher process.[32]
Ilmenite ores can also be converted to liquid iron and a titanium-rich slag using a smelting process.[33]
Ilmenite ore is used as a flux by steelmakers to line blast furnace hearth refractory.[34]
Ilmenite can be used to produce ferrotitanium via an aluminothermic reduction.[35]
Feedstock production
| Feedstock | TiO 2 Content |
Process |
|---|---|---|
| (%) | ||
| Ore | <55 | Sulfate |
| Ore | >55 | Chloride |
| Ore | <50 | Smelting (slag) |
| Synthetic rutile | 88–95 | Chloride |
| Chloride slag | 85–95 | Chloride |
| Sulfate slag | 80 | Sulfate |
| Year | 2011 | 2012–13 |
|---|---|---|
| Country | USGS | Projected |
| Australia | 1,300 | 247 |
| South Africa | 1,161 | 190 |
| Mozambique | 516 | 250 |
| Canada | 700 | |
| India | 574 | |
| China | 500 | |
| Vietnam | 490 | |
| Ukraine | 357 | |
| Senegal | - | 330 |
| Norway | 300 | |
| United States | 300 | |
| Madagascar | 288 | |
| Kenya | - | 246 |
| Sri Lanka | 62 | |
| Sierra Leone | 60 | |
| Brazil | 48 | |
| Other countries | 37 | |
| Total world | ~6,700 | ~1,250 |
Most ilmenite is recovered from heavy mineral sands ore deposits, where the mineral is concentrated as a placer deposit and weathering reduces its iron content, increasing the percentage of titanium. However, ilmenite can also be recovered from "hard rock" titanium ore sources, such as ultramafic to mafic layered intrusions or anorthosite massifs. The ilmenite in layered intrusions is sometimes abundant, but it contains considerable intergrowths of magnetite that reduce its ore grade. Ilmenite from anorthosite massifs often contain large amounts of calcium or magnesium that render it unsuitable for the chloride process.[39]
The proven reserves of ilmenite and rutile ore are estimated at between 423 and 600 million tonnes titanium dioxide. The largest ilmenite deposits are in South Africa, India, the United States, Canada, Norway, Australia, Ukraine, Russia and Kazakhstan. Additional deposits are found in Bangladesh, Chile, Mexico and New Zealand.[40]
Australia was the world's largest ilmenite ore producer in 2011, with about 1.3 million tonnes of production, followed by South Africa, Canada, Mozambique, India, China, Vietnam, Ukraine, Norway, Madagascar and United States.
The top four ilmenite and rutile feedstock producers in 2010 were Rio Tinto Group, Iluka Resources, Exxaro and Kenmare Resources, which collectively accounted for more than 60% of world's supplies.[41]
The world's two largest open cast ilmenite mines are:
- The Tellnes mine located in Sokndal, Norway, and run by Titania AS (owned by Kronos Worldwide Inc.) with 0.55 Mtpa capacity and 57 Mt contained TiO
2 reserves. - The Rio Tinto Group's Lac Tio mine located near Havre Saint-Pierre, Quebec in Canada with a 3 Mtpa capacity and 52 Mt reserves.[42]
Major mineral sands based ilmenite mining operations include:
- Richards Bay Minerals in South Africa, majority-owned by the Rio Tinto Group.
- Kenmare Resources' Moma mine in Mozambique.
- Iluka Resources' mining operations in Australia including Murray Basin, Eneabba and Capel.
- The Kerala Minerals & Metals Ltd (KMML), Indian Rare Earths (IRE), VV Mineral mines in India.
- TiZir Ltd.'s Grande Cote mine in Senegal[43]
- QIT Madagascar Minerals mine, majority-owned by the Rio Tinto Group, which began production in 2009 and is expected to produce 0.75 Mtpa of ilmenite, potentially expanding to 2 Mtpa in future phases.
Attractive major potential ilmenite deposits include:
- The Karhujupukka magnetite-ilmenite deposit in Kolari, northern Finland with around 5 Mt reserves and ore containing about 6.2% titanium.
- The Balla Balla magnetite-iron-titanium-vanadium ore deposit in the Pilbara of Western Australia, which contains 456 million tonnes of cumulate ore horizon grading 45% Fe, 13.7% TiO
2 and 0.64% V
2O
5, one of the richest magnetite-ilmenite ore bodies in Australia[44] - The Coburn, WIM 50, Douglas, Pooncarie mineral sands deposits in Australia.
- The Magpie titano-magnetite (iron-titanium-vanadium-chrome) deposits in eastern Quebec of Canada with about 1 billion tonnes containing about 43% Fe, 12% TiO2, 0.4% V2O5, and 2.2% Cr2O3.
- The Longnose deposit in Northeast Minnesota is considered to be "the largest and richest ilmenite deposit in North America."[45]
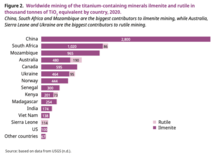
In 2020, China has by far the highest titanium mining activity. About 35 percent of the world’s ilmenite is mined in China, representing 33 percent of total titanium mineral mining (including ilmenite and rutile). South Africa and Mozambique are also important contributors, representing 13 percent and 12 percent of worldwide ilmenite mining, respectively. Australia represents 6 percent of the total ilmenite mining and 31 percent of rutile mining. Sierra Leone and Ukraine are also big contributors to rutile mining.[30]
China is the biggest producer of titanium dioxide, followed by the United States and Germany. China is also the leader in the production of titanium metal, but Japan, the Russian Federation and Kazakhstan have emerged as important contributors to this field.
Patenting activities

Patenting activity related to titanium dioxide production from ilmenite is rapidly increasing.[30] Between 2002 and 2022, there have been 459 patent families that describe the production of titanium dioxide from ilmenite, and this number is growing rapidly. The majority of these patents describe pre-treatment processes, such as using smelting and magnetic separation to increase titanium concentration in low-grade ores, leading to titanium concentrates or slags. Other patents describe processes to obtain titanium dioxide, either by a direct hydrometallurgical process or through two industrially exploited processes, the sulfate process and the chloride process. Acid leaching might be used either as a pre-treatment or as part of a hydrometallurgical process to directly obtain titanium dioxide or synthetic rutile (>90 percent titanium dioxide, TiO2). The sulfate process represents 40 percent of the world’s titanium dioxide production and is protected in 23 percent of patent families. The chloride process is only mentioned in 8 percent of patent families, although it provides 60 percent of the worldwide industrial production of titanium dioxide.[30]
Key contributors to patents on the production of titanium dioxide are companies from China, Australia and the United States, reflecting the major contribution of these countries to industrial production. Chinese companies Pangang and Lomon Billions Groups are the main contributors and hold diversified patent portfolios covering both pre-treatment and the processes leading to a final product.
In comparison, patenting activity related to titanium metal production from ilmenite remains stable.[30] Between 2002 and 2022, there have been 92 patent families that describe the production of titanium metal from ilmenite, and this number has remained quite steady. These patents describe the production of titanium metal starting from mineral ores, such as ilmenite, and from titanium dioxide (TiO2) and titanium tetrachloride (TiCl4), a chemical obtained as an intermediate in the chloride process. The starting materials are purified if needed, and then converted to titanium metal by a chemical reduction process using a reducing agent. Processes mainly differ in regard to the reducing agent used to transform the starting material into titanium metal: magnesium is the most frequently cited reducing agent and the most exploited in industrial production.
Key players in the field are Japanese companies, in particular Toho Titanium and Osaka Titanium Technologies, both focusing on reduction using magnesium. Pangang also contributes to titanium metal production and holds patents describing reduction by molten salt electrolysis.[30]
Lunar ilmenite
Ilmenite has been found in lunar samples, particularly in high-Ti lunar mare basalts common from Apollo 11 and Apollo 17 sites, and on average, constitutes up to 5% of lunar meteorites.[46] Ilmenite has been targeted for ISRU water and oxygen extraction due to a simplistic reduction reaction which occurs with CO and H2 buffers.[47][48][49]
Sources
![]() This article incorporates text from a free content work. Licensed under CC-BY. Text taken from Production of titanium and titanium dioxide from ilmenite and related applications, WIPO.
This article incorporates text from a free content work. Licensed under CC-BY. Text taken from Production of titanium and titanium dioxide from ilmenite and related applications, WIPO.
References
- ^ Warr, L.N. (2021). "IMA–CNMNC approved mineral symbols". Mineralogical Magazine. 85 (3): 291–320. Bibcode:2021MinM...85..291W. doi:10.1180/mgm.2021.43. S2CID 235729616.
- ^ Barthelmy, David (2014). "Ilmenite Mineral Data". Mineralogy Database. Webmineral.com. Retrieved 12 February 2022.
- ^ a b c d e Anthony, John W.; Bideaux, Richard A.; Bladh, Kenneth W.; Nichols, Monte C. (eds.). "Ilmenite". Handbook of Mineralogy (PDF). Chantilly, VA, USA: Mineralogical Society of America. Retrieved 12 February 2022.
- ^ ilmenite, Mindat.org
- ^ Heinz Sibum, Volker Günther, Oskar Roidl, Fathi Habashi, Hans Uwe Wolf, "Titanium, Titanium Alloys, and Titanium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a27_095
- ^ "Sachtleben RDI-S" (PDF). Archived from the original (PDF) on 25 December 2018. Retrieved 25 December 2018.
- ^ "Products". Mineral Commodities Ltd. Retrieved 8 August 2016.
- ^ a b c d e f Klein, Cornelis; Hurlbut, Cornelius S. Jr. (1993). Manual of mineralogy : (after James D. Dana) (21st ed.). New York: Wiley. pp. 380–381. ISBN 047157452X.
- ^ a b c Sinkankas, John (1964). Mineralogy for amateurs. Princeton, N.J.: Van Nostrand. pp. 328–329. ISBN 0442276249.
- ^ Nesse, William D. (2000). Introduction to mineralogy. New York: Oxford University Press. pp. 366–367. ISBN 9780195106916.
- ^ Gregor, William (1791) "Beobachtungen und Versuche über den Menakanit, einen in Cornwall gefundenen magnetischen Sand" (Observations and experiments regarding menaccanite [i.e., ilmenite], a magnetic sand found in Cornwall), Chemische Annalen …, 1, pp. 40–54, 103–119.
- ^ Emsley, John (2001). "Titanium". Nature's Building Blocks: An A-Z Guide to the Elements. Oxford, England, UK: Oxford University Press. ISBN 978-0-19-850340-8.
- ^ Woodford, Chris (2003). Titanium. New York: Benchmark Books. p. 7. ISBN 9780761414612. Retrieved 22 February 2022.
- ^ Habashi, Fathi (January 2001). "Historical Introduction to Refractory Metals". Mineral Processing and Extractive Metallurgy Review. 22 (1): 25–53. Bibcode:2001MPEMR..22...25H. doi:10.1080/08827509808962488. S2CID 100370649.
- ^ Wyatt, Bruce A.; Baumgartner, Mike; Anckar, Eva; Grutter, Herman (September 2004). "Compositional classification of "kimberlitic" and "non-kimberlitic" ilmenite". Lithos. 77 (1–4): 819–840. Bibcode:2004Litho..77..819W. doi:10.1016/j.lithos.2004.04.025. S2CID 140539776.
- ^ a b Sasaki, Kazuhiro; Nakashima, Kazuo; Kanisawa, Satoshi (15 July 2003). "Pyrophanite and high Mn ilmenite discovered in the Cretaceous Tono pluton, NE Japan". Neues Jahrbuch für Mineralogie - Monatshefte. 2003 (7): 302–320. doi:10.1127/0028-3649/2003/2003-0302.
- ^ Weibel, Rikke; Friis, Henrik (2007). "Chapter 10 Alteration of Opaque Heavy Minerals as a Reflection of the Geochemical Conditions in Depositional and Diagenetic Environments". Developments in Sedimentology. 58: 277–303. doi:10.1016/S0070-4571(07)58010-6. ISBN 9780444517531.
- ^ a b Buddington, A. F.; Lindsley, D. H. (1 January 1964). "Iron-Titanium Oxide Minerals and Synthetic Equivalents". Journal of Petrology. 5 (2): 310–357. doi:10.1093/petrology/5.2.310.
- ^ a b Murphy, P.; Frick, L. (2006). "Titanium". In Kogel, J. (ed.). Industrial minerals & rocks: commodities, markets, and uses. SME. pp. 987–1003. ISBN 9780873352338. Retrieved 21 February 2022.
- ^ Mücke, A.; Bhadra Chaudhuri, J.N. (February 1991). "The continuous alteration of ilmenite through pseudorutile to leucoxene". Ore Geology Reviews. 6 (1): 25–44. Bibcode:1991OGRv....6...25M. doi:10.1016/0169-1368(91)90030-B.
- ^ Van Gosen, Bradley S.; Fey, David L.; Shah, Anjana K.; Verplanck, Philip L.; Hoefen, Todd M. (2014). "Deposit model for heavy-mineral sands in coastal environments". U.S. Geological Survey Scientific Investigations Report. Scientific Investigations Report. 201--5070-L. doi:10.3133/sir20105070L.
- ^ Wilson, J.R.; Robins, B.; Nielsen, F.M.; Duchesne, J.C.; Vander Auwera, J. (1996). "The Bjerkreim-Sokndal Layered Intrusion, Southwest Norway". Developments in Petrology. 15: 231–255. doi:10.1016/S0167-2894(96)80009-1. hdl:2268/550. ISBN 9780444817686.
- ^ Charlier, Bernard; Sakoma, Emmanuel; Sauvé, Martin; Stanaway, Kerry; Auwera, Jacqueline Vander; Duchesne, Jean-Clair (March 2008). "The Grader layered intrusion (Havre-Saint-Pierre Anorthosite, Quebec) and genesis of nelsonite and other Fe–Ti–P ores". Lithos. 101 (3–4): 359–378. Bibcode:2008Litho.101..359C. doi:10.1016/j.lithos.2007.08.004.
- ^ Dawson, J.Barry; Smith, Joseph V. (February 1977). "The MARID (mica-amphibole-rutile-ilmenite-diopside) suite of xenoliths in kimberlite". Geochimica et Cosmochimica Acta. 41 (2): 309–323. Bibcode:1977GeCoA..41..309D. doi:10.1016/0016-7037(77)90239-3.
- ^ Cordeiro, Pedro F.O.; Brod, José A.; Dantas, Elton L.; Barbosa, Elisa S.R. (August 2010). "Mineral chemistry, isotope geochemistry and petrogenesis of niobium-rich rocks from the Catalão I carbonatite-phoscorite complex, Central Brazil". Lithos. 118 (3–4): 223–237. Bibcode:2010Litho.118..223C. doi:10.1016/j.lithos.2010.04.007.
- ^ "Industry Fundamentals". Mineral Commodities Ltd. Archived from the original on 7 October 2016. Retrieved 8 August 2016.
- ^ Kroll, W (1940). "The production of ductile titanium". Transactions of the Electrochemical Society. 78: 35–47. doi:10.1149/1.3071290.
- ^ Seki, Ichiro (2017). "Reduction of titanium dioxide to metallic titanium by nitridization and thermal decomposition". Materials Transactions. 58 (3): 361–366. doi:10.2320/matertrans.MK201601.
- ^ "Titanium Dioxide Chemical Economics Handbook".
- ^ a b c d e f World Intellectual Property Organization. (2023). "Patent Landscape Report". WIPO. Patent Landscape Reports. doi:10.34667/tind.47029. Retrieved 19 October 2023.
- ^ Völz, Hans G.; et al. (2006). "Pigments, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a20_243.pub2. ISBN 978-3527306732.
- ^ Welham, N.J. (December 1996). "A parametric study of the mechanically activated carbothermic reduction of ilmenite". Minerals Engineering. 9 (12): 1189–1200. Bibcode:1996MiEng...9.1189W. doi:10.1016/S0892-6875(96)00115-X.
- ^ Pistorius, P.C. (January 2008), "Ilmenite smelting: the basics" (PDF), The Journal of the South African Institute of Mining and Metallurgy, 108
- ^ "Rio Tinto, Fer et Titane - Products". Rio Tinto Group. Archived from the original on 6 May 2015. Retrieved 19 August 2012.
- ^ Gasik, Michael, ed. (2013). Handbook of Ferroalloys: Theory and Technology. London: Elsevier. p. 429. ISBN 978-0-08-097753-9.
- ^ Hayes, Tony (2011), Titanium Dioxide: A Shining Future Ahead (PDF), Euro Pacific Canada, p. 5, retrieved 16 August 2012[dead link]
- ^ Hayes 2011, p. 5.
- ^ USGS 2012 Survey, p. 174
- ^ Murphy, Philip; Frick, Louise (2006). "Titanium". In Barker, James M.; Kogel, Jessica Elzea; Trivedi, Nikhil C.; Krukowski, Stanley T. (eds.). Industrial minerals & rocks : commodities, markets, and uses (7th ed.). Littleton, Colo.: Society for Mining, Metallurgy, and Exploration. pp. 990–991. ISBN 9780873352338. Retrieved 23 February 2022.
- ^ Güther, V.; Sibum, H.; Roidl, O.; Habashi, F.; Wolf, H (2005). "Titanium, Titanium Alloys, and Titanium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Wiley InterScience. ISBN 978-3-527-30673-2.
- ^ Hayes 2011, p. 3.
- ^ "Lac Tio Mine". InfoMine. Retrieved 16 August 2012.
- ^ "TiZir Limited". Mineral Deposits Limited. Archived from the original on 18 August 2012. Retrieved 16 August 2012.
- ^ "Vanadium - AIMR 2011 - Australian Mines Atlas".
- ^ Kraker, Dan. "Titanium Range? Breakthrough could lead to new kind of mining in NE Minn". Retrieved 31 May 2017.
- ^ Korotev, Randy. 2005 "Lunar geochemistry as told by lunar meteorites." Geochemistry. Vol 65. Pages 297–346. https://doi.org/10.1016/j.chemer.2005.07.001
- ^ Schluter & Cowley. "Review of techniques for In-Situ oxygen extraction on the moon." Planetary and Space Science. Vol 181. https://doi.org/10.1016/j.pss.2019.104753
- ^ Perreault & Patience. "Ilmenite–CO reduction kinetics." Fuel. Vol 165. Pages 166-172. https://doi.org/10.1016/j.fuel.2015.10.066
- ^ Muscatello, Tony. 2017. "Oxygen Extraction from Minerals" Presentation, NASA KSC Applied Chem lab. https://ntrs.nasa.gov/api/citations/20170001458/downloads/20170001458.pdf




