Enantiomer
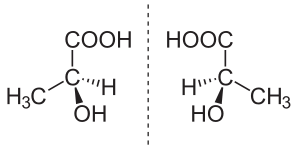
In chemistry, an enantiomer (/ɪˈnænti.əmər, ɛ-, -oʊ-/[1] ih-NAN-tee-ə-mər; from Ancient Greek ἐναντίος (enantíos) 'opposite', and μέρος (méros) 'part') – also called optical isomer,[2] antipode,[3] or optical antipode[4] – is one of a pair of molecular entities which are mirror images of each other and non-superposable. Enantiomers of each other are much like one's right and left hands; without mirroring one of them, hands cannot be superposed onto each other.[5] It is solely a relationship of chirality and the permanent three-dimensional relationships among molecules or other chemical structures: no amount of re-orientiation of a molecule as a whole or conformational change converts one chemical into its enantiomer. Chemical structures with chirality rotate plane-polarized light.[6] A mixture of equal amounts of each enantiomer, a racemic mixture or a racemate, does not rotate light.[7][8][9]
Stereoisomers include both enantiomers and diastereomers. Diastereomers, like enantiomers, share the same molecular formula and are also nonsuperposable onto each other; however, they are not mirror images of each other.[10]
Naming conventions
There are three common naming conventions for specifying one of the two enantiomers (the absolute configuration) of a given chiral molecule: the R/S system is based on the geometry of the molecule; the (+)- and (−)- system (also written using the obsolete equivalents d- and l-) is based on its optical rotation properties; and the D/L system is based on the molecule's relationship to enantiomers of glyceraldehyde.
The R/S system is based on the molecule's geometry with respect to a chiral center.[11] The R/S system is assigned to a molecule based on the priority rules assigned by Cahn–Ingold–Prelog priority rules, in which the group or atom with the largest atomic number is assigned the highest priority and the group or atom with the smallest atomic number is assigned the lowest priority.
The (+) or (−) symbol is used to specify a molecule's optical rotation — the direction in which the polarization of light rotates as it passes through a solution containing the molecule.[12] When a molecule is denoted dextrorotatory, it rotates the plane of polarized light clockwise and can also be denoted as (+).[11] When it is denoted as levorotatory, it rotates the plane of polarized light counterclockwise and can also be denoted as (−).[11]
The Latin words for left are laevus and sinister, and the word for right is dexter (or rectus in the sense of correct or virtuous). The English word right is a cognate of rectus. This is the origin of the D/L and R/S notations, and the employment of prefixes levo- and dextro- in common names.
The prefix ar-, from the Latin recto (right), is applied to the right-handed version; es-, from the Latin sinister (left), to the left-handed molecule. Example: ketamine, arketamine, esketamine.[13]
Chirality centers
The asymmetric atom is called a chirality center,[14][15] a type of stereocenter. A chirality center is also called a chiral center[16][17][18] or an asymmetric center.[19] Some sources use the terms stereocenter, stereogenic center, stereogenic atom or stereogen to refer exclusively to a chirality center,[16][18][20] while others use the terms more broadly to refer also to centers that result in diastereomers (stereoisomers that are not enantiomers).[15][21][22]
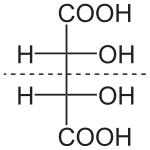
Compounds that contain exactly one (or any odd number) of asymmetric atoms are always chiral. However, compounds that contain an even number of asymmetric atoms sometimes lack chirality because they are arranged in mirror-symmetric pairs, and are known as meso compounds. For instance, meso tartaric acid (shown on the right) has two asymmetric carbon atoms, but it does not exhibit enantiomerism because there is a mirror symmetry plane. Conversely, there exist forms of chirality that do not require asymmetric atoms, such as axial, planar, and helical chirality.[16]: pg. 3
Even though a chiral molecule lacks reflection (Cs) and rotoreflection symmetries (S2n), it can have other molecular symmetries, and its symmetry is described by one of the chiral point groups: Cn, Dn, T, O, or I. For example, hydrogen peroxide is chiral and has C2 (two-fold rotational) symmetry. A common chiral case is the point group C1, meaning no symmetries, which is the case for lactic acid.
Examples

An example of such an enantiomer is the sedative thalidomide, which was sold in a number of countries around the world from 1957 until 1961. It was withdrawn from the market when it was found to cause birth defects. One enantiomer caused the desirable sedative effects, while the other, unavoidably[23] present in equal quantities, caused birth defects.[24]
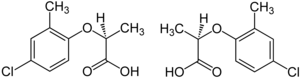
The herbicide mecoprop is a racemic mixture, with the (R)-(+)-enantiomer ("Mecoprop-P", "Duplosan KV") possessing the herbicidal activity.[25]
Another example is the antidepressant drugs escitalopram and citalopram. Citalopram is a racemate [1:1 mixture of (S)-citalopram and (R)-citalopram]; escitalopram [(S)-citalopram] is a pure enantiomer. The dosages for escitalopram are typically 1/2 of those for citalopram. Here, (S)-citalopram is called a chiral switch of Citalopram.
Chiral drugs
Enantiopure compounds consist of only one of the two enantiomers. Enantiopurity is of practical importance since such compositions have improved therapeutic efficacy.[26] The switch from a racemic drug to an enantiopure drug is called a chiral switch. In many cases, the enantiomers have distinct effects. One case is that of Propoxyphene. The enantiomeric pair of propoxyphene is separately sold by Eli Lilly and company. One of the partners is dextropropoxyphene, an analgesic agent (Darvon) and the other is called levopropoxyphene, an effective antitussive (Novrad).[27][28] It is interesting to note that the trade names of the drugs, DARVON and NOVRAD, also reflect the chemical mirror-image relationship. In other cases, there may be no clinical benefit to the patient. In some jurisdictions, single-enantiomer drugs are separately patentable from the racemic mixture.[29] It is possible that only one of the enantiomers is active. Or, it may be that both are active, in which case separating the mixture has no objective benefits, but extends the drug's patentability.[30]
Enantioselective preparations
In the absence of an effective enantiomeric environment (precursor, chiral catalyst, or kinetic resolution), separation of a racemic mixture into its enantiomeric components is impossible, although certain racemic mixtures spontaneously crystallize in the form of a racemic conglomerate, in which crystals of the enantiomers are physically segregated and may be separated mechanically. However, most racemates form crystals containing both enantiomers in a 1:1 ratio.
In his pioneering work, Louis Pasteur was able to isolate the isomers of sodium ammonium tartrate because the individual enantiomers crystallize separately from solution. To be sure, equal amounts of the enantiomorphic crystals are produced, but the two kinds of crystals can be separated with tweezers. This behavior is unusual. A less common method is by enantiomer self-disproportionation.
The second strategy is asymmetric synthesis: the use of various techniques to prepare the desired compound in high enantiomeric excess. Techniques encompassed include the use of chiral starting materials (chiral pool synthesis), the use of chiral auxiliaries and chiral catalysts, and the application of asymmetric induction. The use of enzymes (biocatalysis) may also produce the desired compound.
A third strategy is Enantioconvergent synthesis, the synthesis of one enantiomer from a racemic precursor, utilizing both enantiomers. By making use of a chiral catalyist, both enantiomers of the reactant result in a single enantiomer of product.[31]
Enantiomers may not be isolable if there is an accessible pathway for racemization (interconversion between enantiomorphs to yield a racemic mixture) at a given temperature and timescale. For example, amines with three distinct substituents are chiral, but with few exceptions (e.g. substituted N-chloroaziridines), they rapidly undergo "umbrella inversion" at room temperature, leading to racemization. If the racemization is fast enough, the molecule can often be treated as an achiral, averaged structure.
Parity violation
For all intents and purposes, each enantiomer in a pair has the same energy. However, theoretical physics predicts that due to parity violation of the weak nuclear force (the only force in nature that can "tell left from right"), there is actually a minute difference in energy between enantiomers (on the order of 10−12 eV or 10−10 kJ/mol or less) due to the weak neutral current mechanism. This difference in energy is far smaller than energy changes caused by even small changes in molecular conformation, and far too small to measure by current technology, and is therefore chemically inconsequential.[17][32][33] In the sense used by particle physicists, the "true" enantiomer of a molecule, which has exactly the same mass-energy content as the original molecule, is a mirror-image that is also built from antimatter (antiprotons, antineutrons, and positrons).[17] Throughout this article, "enantiomer" is used only in the chemical sense of compounds of ordinary matter that are not superposable on their mirror image.
Quasi-enantiomers
Quasi-enantiomers are molecular species that are not strictly enantiomers, but behave as if they are. In quasi-enantiomers majority of the molecule is reflected; however, an atom or group within the molecule is changed to a similar atom or group.[34] Quasi-enantiomers can also be defined as molecules that have the potential to become enantiomers if an atom or group in the molecule is replaced.[35] An example of quasi-enantiomers would (S)-bromobutane and (R)-iodobutane. Under normal conditions the enantiomers for (S)-bromobutane and (R)-iodobutane would (R)-bromobutane and (S)-iodobutane respectively. Quasi-enantiomers would also produce quasi-racemates, which are similar to normal racemates (see Racemic mixture) in that they form an equal mixture of quasi-enantiomers.[34]
Though not considered actual enantiomers, the naming convention for quasi-enantiomers also follows the same trend as enantiomers when looking at (R) and (S) configurations - which are considered from a geometrical basis (see Cahn–Ingold–Prelog priority rules).
Quasi-enantiomers have applications in parallel kinetic resolution.[36]
See also
References
- ^ "Enantiomer: Definition & Meaning". Dictionary.com. Retrieved 2024-04-26.
- ^ Chemistry (IUPAC), The International Union of Pure and Applied. "IUPAC - optical isomers (O04308)". goldbook.iupac.org. doi:10.1351/goldbook.O04308. Retrieved 2022-11-17.
- ^ Chemistry (IUPAC), The International Union of Pure and Applied. "IUPAC - antipodes (A00403)". goldbook.iupac.org. doi:10.1351/goldbook.A00403. Retrieved 2022-11-17.
- ^ Chemistry (IUPAC), The International Union of Pure and Applied. "IUPAC - optical antipodes (O04304)". goldbook.iupac.org. doi:10.1351/goldbook.O04304. Retrieved 2022-11-17.
- ^ McConathy, Jonathan; Owens, Michael J. (2003). "Stereochemistry in Drug Action". Primary Care Companion to the Journal of Clinical Psychiatry. 5 (2): 70–73. doi:10.4088/pcc.v05n0202. ISSN 1523-5998. PMC 353039. PMID 15156233.
- ^ "Chirality and Optical Activity". chemed.chem.purdue.edu. Retrieved 2022-11-17.
- ^ Chemistry (IUPAC), The International Union of Pure and Applied. "IUPAC - racemic (R05026)". goldbook.iupac.org. doi:10.1351/goldbook.R05026. Retrieved 2022-11-17.
- ^ Chemistry (IUPAC), The International Union of Pure and Applied. "IUPAC - racemate (R05025)". goldbook.iupac.org. doi:10.1351/goldbook.R05025. Retrieved 2022-11-17.
- ^ Weber, Erin. "Library Guides: CHEM 221: Stereochemistry / Isomerism". libraryguides.salisbury.edu. Retrieved 2022-11-17.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1
- ^ a b c Brewster, James H. (December 1986). "The distinction of diastereomers in the Cahn-Ingold-Prelog (RS) notation". The Journal of Organic Chemistry. 51 (25): 4751–4753. doi:10.1021/jo00375a001. ISSN 0022-3263.
- ^ Caldwell, John; Wainer, Irving W. (December 2001). "Stereochemistry: definitions and a note on nomenclature". Human Psychopharmacology: Clinical and Experimental. 16 (S2): S105–S107. doi:10.1002/hup.334. ISSN 0885-6222. PMID 12404716. S2CID 12367578.
- ^ Long, Aaron S.; Zhang, Audrey D.; Meyer, Caitlin E.; Egilman, Alexander C.; Ross, Joseph S.; Wallach, Joshua D. (May 6, 2021). "Evaluation of Trials Comparing Single-Enantiomer Drugs to Their Racemic Precursors: A Systematic Review". JAMA Network Open. 4 (5): e215731. doi:10.1001/jamanetworkopen.2021.5731. PMC 8103227 – via Silverchair.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "chirality centre". doi:10.1351/goldbook.C01060
- ^ a b Wade, LeRoy G. (2006). "Precision in Stereochemical Terminology". J. Chem. Educ. 83 (12): 1793. Bibcode:2006JChEd..83.1793W. doi:10.1021/ed083p1793. ISSN 0021-9584.
- ^ a b c Karras, Manfred (2018). "Synthesis of Enantiomerically Pure Helical Aromatics Such As NHC Ligands and Their Use in Asymmetric Catalysis (PhD). Charles University. Retrieved 6 August 2021.
- ^ a b c Eliel, Ernest L.; Wilen, Samuel H.; Mander, Lewis N. (1994). Stereochemistry of organic compounds. New York: Wiley. ISBN 0471016705. OCLC 27642721.
- ^ a b Clayden, Jonathan; Greeves, Nick; Warren, Stuart G. (2012). Organic chemistry. Oxford: Oxford University Press. ISBN 978-0-19-927029-3. OCLC 761379371.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "asymmetric centre". doi:10.1351/goldbook.A00480
- ^ Clark, Andrew; Kitson, Russell R. A.; Mistry, Nimesh; Taylor, Paul; Taylor, Matthew; Lloyd, Michael; Akamune, Caroline (2021). Introduction to stereochemistry. Cambridge, UK. ISBN 978-1-78801-315-4. OCLC 1180250839.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "stereogenic unit (stereogen/stereoelement)". doi:10.1351/goldbook.S05980
- ^ Mislow, Kurt; Siegel, Jay (1984). "Stereoisomerism and local chirality". J. Am. Chem. Soc. 106 (11): 3319–3328. doi:10.1021/ja00323a043. ISSN 0002-7863.
- ^ Knoche, B; Blaschke, G (1994). "Investigations on the in vitro racemization of thalidomide by high-performance liquid chromatography". Journal of Chromatography A. 666 (1–2): 235–240. doi:10.1016/0021-9673(94)80385-4.
- ^ Voet, Donald; Voet, Judith G.; Pratt, Charlotte W. (2006). Fundamentals of Biochemistry. p. 89. ISBN 0-471-21495-7.
- ^ G. Smith; C. H. L. Kennard; A. H. White; P. G. Hodgson (April 1980). "(±)-2-(4-Chloro-2-methylphenoxy)propionic acid (mecoprop)". Acta Crystallogr. B. 36 (4): 992–994. doi:10.1107/S0567740880005134.
- ^ Ariëns, Everardus J. (1986). "Stereochemistry: A source of problems in medicinal chemistry". Medicinal Research Reviews. 6 (4): 451–466. doi:10.1002/med.2610060404. ISSN 0198-6325. PMID 3534485. S2CID 36115871.
- ^ Drayer, Dennis E (1986). "Pharmacodynamic and pharmacokinetic differences between drug enantiomers in humans: An overview". Clinical Pharmacology and Therapeutics. 40 (2): 125–133. doi:10.1038/clpt.1986.150. ISSN 0009-9236. PMID 3731675. S2CID 33537650.
- ^ Ariens, E.J (1989). Chiral Separations by HPLC. Chichester: Ellis Horwwod. pp. 31–68.
- ^ "European Medicines Agency - - Sepracor Pharmaceuticals Ltd withdraws its marketing authorisation application for Lunivia (eszopiclone)". www.ema.europa.eu. 17 September 2018. Archived from the original on 1 December 2017. Retrieved 14 February 2011.
- ^ Merrill Goozner (2004). The $800 Million Pill: The Truth Behind the Cost of New Drugs (excerpt). University of California Press. ISBN 0-520-23945-8.
- ^ Mohr, J.T.; Moore, J.T.; Stoltz, B.M. (2016). "Enantioconvergent catalysis". Beilstein J. Org. Chem. 12: 2038–2045. doi:10.3762/bjoc.12.192. PMC 5082454. PMID 27829909. Retrieved 4 August 2021.
- ^ Albert, Guijarro (2008). The origin of chirality in the molecules of life: a revision from awareness to the current theories and perspectives of this unsolved problem. Yus, Miguel. Cambridge, UK: Royal Society of Chemistry. ISBN 9781847558756. OCLC 319518566.
- ^ Stickler, Benjamin A.; Diekmann, Mira; Berger, Robert; Wang, Daqing (2021-09-14). "Enantiomer Superpositions from Matter-Wave Interference of Chiral Molecules". Physical Review X. 11 (3): 031056. arXiv:2102.06124. Bibcode:2021PhRvX..11c1056S. doi:10.1103/PhysRevX.11.031056. ISSN 2160-3308. S2CID 231879820.
- ^ a b Zhang, Qisheng; Rivkin, Alexey; Curran, Dennis P. (2002-05-01). "Quasiracemic Synthesis: Concepts and Implementation with a Fluorous Tagging Strategy to Make Both Enantiomers of Pyridovericin and Mappicine". Journal of the American Chemical Society. 124 (20): 5774–5781. doi:10.1021/ja025606x. ISSN 0002-7863. PMID 12010052.
- ^ Zhang, Qisheng; Curran, Dennis P. (2005-08-19). "Quasienantiomers and Quasiracemates: New Tools for Identification, Analysis, Separation, and Synthesis of Enantiomers". Chemistry - A European Journal. 11 (17): 4866–4880. doi:10.1002/chem.200500076. ISSN 0947-6539. PMID 15915521.
- ^ G.S. Coumbarides, M. Dingjan, J. Eames, A. Flinn, J. Northen and Y. Yohannes, Tetrahedron Lett. 46 (2005), p. 2897er
External links
 Media related to Enantiomers at Wikimedia Commons
Media related to Enantiomers at Wikimedia Commons- chemwiki:stereoisomerism
