Diketopyrrolopyrrole dye
Diketopyrrolopyrroles (DPPs) are organic dyes and pigments based on the heterocyclic dilactam 2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione,[1][2][3] widely used in optoelectronics. DPPs were initially used as pigments in the painting industry (e.g. in automotive paints) due to their high resistance to photodegradation. More recently, DPP derivatives have been also investigated as promising fluorescent dyes for bioimaging applications,[4] as well as components of materials for use in organic electronics.[5][6][7][8][9][10][11][3][12]
Structure
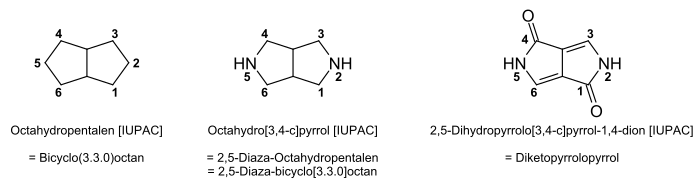
DPP dyes are based on the bicyclic heterocyclic compound diketopyrrolopyrrole. 2,5-Dihydropyrrolo[3,4-c]pyrrole-1,4-dione is a basic body of Diketopyrrolopyrrole dye. DPP pigments are an important class of high-performance pigments used in inks, paints and plastic. More recently their optical-electronic performance was discovered for photovoltaic applications and related uses.[13]
Optical properties
DPP has high stability to heat but not very well to acid and base. The alkylation of amide groups improves the solubility. The modified DDP shows high fluorescence and the emission wavelength is adjustable by change the aromatic group at 3 and 6 positions. Both electron donor group, such as thiophene, and expanding of the conjugated system increase the emission wavelength. DPP is also developed as a red emitting group in pure polymer light emitting diodes.[14][15][16][17] Some DPP derivatives show aggregation induced emission[18] and were used as effective fluorescent sensors, in organic solar cells.[19][20][21][22][23][24][25][26][27][28]
References
- ^ Wallquist, O.; Lenz, R. (2002-03-01). "DPP chemistry — continuous innovation". Surface Coatings International Part B: Coatings Transactions. 85 (1): 19–26. doi:10.1007/BF02699738. ISSN 1476-4865. S2CID 98773045.
- ^ Grzybowski, Marek; Gryko, Daniel T. (2015). "Diketopyrrolopyrroles: Synthesis, Reactivity, and Optical Properties". Advanced Optical Materials. 3 (3): 280–320. doi:10.1002/adom.201400559. ISSN 2195-1071.
- ^ a b Tian, He; Qu, Sanyin (2012-02-27). "Diketopyrrolopyrrole (DPP)-based materials for organic photovoltaics". Chemical Communications. 48 (25): 3039–3051. doi:10.1039/C2CC17886A. ISSN 1364-548X. PMID 22343975.
- ^ Choi, Dong Hoon; Kaur, Matinder (2014-12-08). "Diketopyrrolopyrrole: brilliant red pigment dye-based fluorescent probes and their applications". Chemical Society Reviews. 44 (1): 58–77. doi:10.1039/C4CS00248B. ISSN 1460-4744. PMID 25186723.
- ^ Khan, Faizal; Jang, Youngwoo; Patil, Yuvraj; Misra, Rajneesh; D'Souza, Francis (2021-09-06). "Photoinduced Charge Separation Prompted Intervalence Charge Transfer in a Bis(thienyl)diketopyrrolopyrrole Bridged Donor‐TCBD Push‐Pull System". Angewandte Chemie International Edition. 60 (37): 20518–20527. doi:10.1002/anie.202108293. ISSN 1433-7851. PMID 34258866. S2CID 235820925.
- ^ Popli, Charu; Jang, Youngwoo; Patil, Yuvraj; Misra, Rajneesh; D'Souza, Francis (2020-11-26). "Formation of Highly Efficient, Long‐Lived Charge Separated States in Star‐Shaped Ferrocene‐Diketopyrrolopyrrole‐Triphenylamine Donor–Acceptor–Donor Conjugates". Chemistry: A European Journal. 26 (66): 15109–15115. doi:10.1002/chem.202002851. ISSN 0947-6539. PMID 32589288. S2CID 220075391.
- ^ Patil, Yuvraj; Popli, Charu; Misra, Rajneesh (2018-02-26). "Near-infrared absorbing tetracyanobutadiene-bridged diketopyrrolopyrroles". New Journal of Chemistry. 42 (5): 3892–3899. doi:10.1039/C7NJ05162J. ISSN 1369-9261.
- ^ Popli, Charu; Patil, Yuvraj; Misra, Rajneesh (2018-12-13). "Design and Synthesis of N -Phenylcarbazole-Substituted Diketopyrrolopyrrole-Based Monomers and Dimers: A Comparative Study: Design and Synthesis of N -Phenylcarbazole-Substituted Diketopyrrolopyrrole-Based Monomers and Dimers: A Comparative Study". European Journal of Organic Chemistry. 2018 (46): 6474–6481. doi:10.1002/ejoc.201801072. S2CID 105757956.
- ^ Patil, Yuvraj; Shinde, Jivan; Misra, Rajneesh (2017-12-01). "Near-infrared absorbing metal functionalized diketopyrrolopyrroles". Journal of Organometallic Chemistry. 852: 48–53. doi:10.1016/j.jorganchem.2017.10.014. ISSN 0022-328X.
- ^ Patil, Yuvraj; Jadhav, Thaksen; Dhokale, Bhausaheb; Misra, Rajneesh (February 2016). "Tuning of the HOMO-LUMO Gap of Symmetrical and Unsymmetrical Ferrocenyl-Substituted Diketopyrrolopyrroles". European Journal of Organic Chemistry. 2016 (4): 733–738. doi:10.1002/ejoc.201501123.
- ^ Patil, Yuvraj; Jadhav, Thaksen; Dhokale, Bhausaheb; Misra, Rajneesh (August 2016). "Design and Synthesis of Low HOMO-LUMO Gap N- Phenylcarbazole-Substituted Diketopyrrolopyrroles". Asian Journal of Organic Chemistry. 5 (8): 1008–1014. doi:10.1002/ajoc.201600194.
- ^ Nielsen, Christian B.; Turbiez, Mathieu; McCulloch, Iain (2013). "Recent Advances in the Development of Semiconducting DPP-Containing Polymers for Transistor Applications" (PDF). Advanced Materials. 25 (13): 1859–1880. doi:10.1002/adma.201201795. hdl:10044/1/14443. ISSN 1521-4095. PMID 23008141. S2CID 10575133.
- ^ Encyclopedia of Interfacial Chemistry: Surface Science and Electrochemistry. Elsevier. 2018-03-29. ISBN 978-0-12-809894-3.
- ^ Jin, Yi; Xu, Yanbin; Qiao, Zhi; Peng, Junbiao; Wang, Baozheng; Cao, Derong (2010). "Enhancement of Electroluminescence Properties of Red Diketopyrrolopyrrole-Doped Copolymers by Oxadiazole and Carbazole Units as Pendants". Polymer. 51 (24): 5726–5733. doi:10.1016/j.polymer.2010.09.046.
- ^ Qiao, Zhi; Peng, Junbiao; Jin, Yi; Liu, Qilin; Weng, Jiena; He, Zhicai; Han, Shaohu; Cao, Derong (2010). "Synthesis and Electroluminescence Properties of Fluorene-co-Diketopyrrolopyrrole-co-Phenothiazine Polymers". Polymer. 51 (5): 1016–1023. doi:10.1016/j.polymer.2009.12.044.
- ^ Xu, Yanbin; Jin, Yi; Lin, Weihong; Peng, Junbiao; Jiang, Huanfeng; Cao, Derong (2010). "Syntheses and Electroluminescence Properties of Red Emitting Copolymers with Different Lengths of Diketopyrrolopyrrole Units". Synthetic Metals. 160 (19–20): 2135–2142. doi:10.1016/j.synthmet.2010.07.044.
- ^ Xu, Yanbin; Jin, Yi; Peng, Junbiao; Wang, Baozheng; Cao, Derong (2010). "Syntheses and Characterization of 4-Octyloxybenzyl Substituted Diketopyrrolopyrrole-Based Red Emitting Copolymers with Low Turn-on Voltage". J. Macromol. Sci., Part A: Pure Appl. Chem. 47 (11): 1059–1068. doi:10.1080/10601325.2010.511091. S2CID 97899350.
- ^ Jin, Yi; Xu, Yanbin; Liu, Yinling; Wang, Lingyun; Jiang, Huanfeng; Li, Xianjie; Cao, Derong (2011). "Synthesis of Novel Diketopyrrolopyrrole-Based Luminophores Showing Crystallization-Induced Emission Enhancement Properties". Dyes Pigm. 90 (3): 311–318. doi:10.1016/j.dyepig.2011.01.005.
- ^ Patil, Yuvraj; Misra, Rajneesh (2019-10-31). "Rational molecular design towards NIR absorption: efficient diketopyrrolopyrrole derivatives for organic solar cells and photothermal therapy". Journal of Materials Chemistry C. 7 (42): 13020–13031. doi:10.1039/C9TC03640G. ISSN 2050-7534. S2CID 203513134.
- ^ Patil, Yuvraj; Misra, Rajneesh (June 2020). "Metal Functionalized Diketopyrrolopyrroles: A Promising Class of Materials for Optoelectronic Applications". The Chemical Record. 20 (6): 596–603. doi:10.1002/tcr.201900061. ISSN 1527-8999. PMID 31833617. S2CID 209340212.
- ^ Patil, Yuvraj; Misra, Rajneesh (September 2018). "Small Molecule Based Non-Fullerene Acceptors: A Comparative Study". The Chemical Record. 18 (9): 1350–1364. doi:10.1002/tcr.201800037. PMID 29888443. S2CID 47012825.
- ^ Patil, Yuvraj; Misra, Rajneesh (2018-02-02). "Diketopyrrolopyrrole-Based and Tetracyano-Bridged Small Molecules for Bulk Heterojunction Organic Solar Cells". Chemistry: An Asian Journal. 13 (3): 220–229. doi:10.1002/asia.201701493. PMID 29219247.
- ^ Patil, Yuvraj; Misra, Rajneesh; Singhal, Rahul; Sharma, Ganesh D. (2017-07-04). "Ferrocene-diketopyrrolopyrrole based non-fullerene acceptors for bulk heterojunction polymer solar cells". Journal of Materials Chemistry A. 5 (26): 13625–13633. doi:10.1039/C7TA03322B. ISSN 2050-7496.
- ^ Patil, Yuvraj; Misra, Rajneesh; Keshtov, M. L.; Sharma, Ganesh D. (2017-02-14). "Small molecule carbazole-based diketopyrrolopyrroles with tetracyanobutadiene acceptor unit as a non-fullerene acceptor for bulk heterojunction organic solar cells". Journal of Materials Chemistry A. 5 (7): 3311–3319. doi:10.1039/C6TA09607G. ISSN 2050-7496.
- ^ Patil, Yuvraj; Misra, Rajneesh; Singh, Manish Kumar; Sharma, Ganesh D. (2017-03-08). "Ferrocene-diketopyrrolopyrrole based small molecule donors for bulk heterojunction solar cells". Physical Chemistry Chemical Physics. 19 (10): 7262–7269. doi:10.1039/C7CP00231A. ISSN 1463-9084. PMID 28239736.
- ^ Patil, Yuvraj; Misra, Rajneesh; Keshtov, M. L.; Sharma, Ganesh D. (2016-03-31). "1,1,4,4-Tetracyanobuta-1,3-diene Substituted Diketopyrrolopyrroles: An Acceptor for Solution Processable Organic Bulk Heterojunction Solar Cells". The Journal of Physical Chemistry C. 120 (12): 6324–6335. doi:10.1021/acs.jpcc.5b12307. ISSN 1932-7447.
- ^ Patil, Yuvraj; Misra, Rajneesh; Sharma, Abhishek; Sharma, Ganesh D. (2016-06-22). "D–A–D–π–D–A–D type diketopyrrolopyrrole based small molecule electron donors for bulk heterojunction organic solar cells". Physical Chemistry Chemical Physics. 18 (25): 16950–16957. doi:10.1039/C6CP02700H. ISSN 1463-9084. PMID 27292157.
- ^ Patil, Yuvraj; Misra, Rajneesh; Chen, F. C.; Sharma, Ganesh D. (2016-08-17). "Small molecule based N-phenyl carbazole substituted diketopyrrolopyrroles as donors for solution-processed bulk heterojunction organic solar cells". Physical Chemistry Chemical Physics. 18 (33): 22999–23005. doi:10.1039/C6CP03767D. ISSN 1463-9084. PMID 27489031.
Further reading
- Chem. Commun., 2012, 48, 3039–3051
