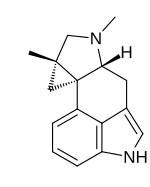
Cycloclavine
| |
| Names | |
|---|---|
| IUPAC name 6,8-Dimethyl-8,10-cycloergoline | |
| Systematic IUPAC name (1aS,3aR,9bS)-1a,3-Dimethyl-1a,2,3,3a,4,6-hexahydro-1H-cyclopropa[c]indolo[4,3-ef]indole | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C16H18N2 | |
| Molar mass | 238.334 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Cycloclavine is an ergot alkaloid. It was first isolated in 1969 from seeds of Ipomoea hildebrandtii vatke.[2] The first total synthesis of (±)-cycloclavine was published in 2008 by Szántay.[3] Further reports came from Wipf and Petronijevic,[4] Cao[5] and Brewer.[6] In 2016, Wipf and McCabe completed an 8-step asymmetric synthesis of (–)-cycloclavine,[7] and in 2018, they expanded this approach toward (+)-cycloclavine and a biological characterization of the binding profile of both enantiomers on 16 brain receptors.[8] Natural (+)- and unnatural (–)-cycloclavine demonstrated significant stereospecificity and unique binding profiles in comparison to LSD (lysergic acid diethylamide), psilocin, and DMT. Differential 5-HT receptor affinities, as well as novel sigma-1 receptor properties, suggest potential future therapeutic opportunities of clavine alkaloid scaffolds.
References
- ^ "KNApSAcK Metabolite Information - C00011221". www.knapsackfamily.com.
- ^ Stauffacher, D; Niklaus, P; Tscherter, H; Weber, H.P; Hofmann, A (1969). "Cycloclavin, ein neues alkaloid aus Ipomoea hildebrandtii vatke—71". Tetrahedron. 25 (24): 5879–87. doi:10.1016/S0040-4020(01)83095-7. PMID 5373534.
- ^ Incze, M.; Dörnyei, G.; Moldvai, I.; Temesvári-Major, E.; Egyed, O.; Szántay, C. (2008). "New routes to clavine-type ergot alkaloids. Part 2: Synthesis of the last, so far not yet synthesized member of the clavine alkaloid family, (±)-cycloclavine". Tetrahedron. 64 (13): 2924–2929. doi:10.1016/j.tet.2008.01.101.
- ^ Petronijevic, F. R.; Wipf, P (2011). "Total Synthesis of (±)-Cycloclavine and (±)-5-epi-Cycloclavine". J. Am. Chem. Soc. 133 (20): 7704–7707. doi:10.1021/ja2026882. PMC 3111057. PMID 21517102.
- ^ Wang, W.; Lu, J.-T.; Zhang, H.-L.; Shi, Z.-F.; Wen, J.; Cao, X.-P (2013). "Formal Synthesis of (±)-Cycloclavine". J. Org. Chem. 79 (1): 122–127. doi:10.1021/jo4023588. PMID 24279324.
- ^ Jabre, N. D.; Watanabe, T.; Brewer, M. (2014). "Formal and total synthesis of (±)-cycloclavine". Tetrahedron Lett. 55 (1): 197–199. doi:10.1016/j.tetlet.2013.10.152. PMC 3915717. PMID 24511164.
- ^ McCabe, Stephanie R. (2017). "Eight-Step Enantioselective Total Synthesis of (−)-Cycloclavine". Angewandte Chemie. 129 (1): 330–333. doi:10.1002/ange.201608820. PMC 5195887. PMID 27860203.
- ^ Wipf, Peter; McCabe, Stephanie (2018-11-20). "Asymmetric Total Synthesis and Biological Evaluation of (+)-Cycloclavine". Synthesis. 51: 213–224. doi:10.1055/s-0037-1610395. ISSN 0039-7881.
