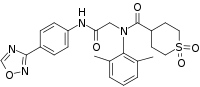
Amenamevir
 | |
| Clinical data | |
|---|---|
| Trade names | Amenalief |
| Other names | ASP-2151, ASP2151 |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H26N4O5S |
| Molar mass | 482.56 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Amenamevir (trade name Amenalief) is an antiviral drug used for the treatment of shingles (herpes zoster).
It acts as an inhibitor of the zoster virus's helicase–primase complex.[1][2] Amenamevir was approved in Japan for the treatment of shingles in 2017.[3]
See also
References
- ^ Kawashima M, Nemoto O, Honda M, Watanabe D, Nakayama J, Imafuku S, et al. (November 2017). "Amenamevir, a novel helicase-primase inhibitor, for treatment of herpes zoster: A randomized, double-blind, valaciclovir-controlled phase 3 study". The Journal of Dermatology. 44 (11): 1219–1227. doi:10.1111/1346-8138.13948. PMC 5697646. PMID 28681394.
- ^ Yajima M, Yamada H, Takemoto M, Daikoku T, Yoshida Y, Long T, et al. (March 2017). "Profile of anti-herpetic action of ASP2151 (amenamevir) as a helicase-primase inhibitor". Antiviral Research. 139: 95–101. doi:10.1016/j.antiviral.2016.12.008. PMID 28027917. S2CID 43813287.
- ^ "Maruho Receives Manufacturing and Marketing Approval for Anti-Herpes Virus Agent "Amenalief® Tab. 200mg" in Japan" (Press release). evaluategroup.com. July 3, 2017.
